Home > Healthcare > Pharmaceuticals > Disease Specific Drugs > Rare Disease Treatment Market
Rare Disease Treatment Market Analysis
- Report ID: GMI3873
- Published Date: Jan 2023
- Report Format: PDF
Rare Disease Treatment Market Analysis
Non-biologics drug segment is set to register over 10% CAGR through 2032, on account of the immunogenicity and minimal side effects associated with such products. These drugs are deemed effective for treating certain conditions including APDS (activated phosphoinositide 3-kinase delta syndrome) and Felty Syndrome. These factors, alongside cost-effectiveness and non-complexity in clinical studies, are projected to augment the consumption of non-biologic medications among rare disease patients.
Rare disease treatment market value from the central nervous system (CNS) segment is expected to surpass USD 38 billion by 2032, given the rising prevalence of disorders that affect nerve cells in the spinal cord and brain, causing loss of muscle control. Based on data from the National Library of Medicine, amyotrophic lateral sclerosis affects around 5 in 100,000 people worldwide. Moreover, the strong focus on the development of precision medicines will further drive the demand for therapies to treat rare diseases.
Pediatric patient segment is anticipated to reach over USD 106 billion by 2032. This is on account of the growing incidence of Byler disease, a rare inherited condition due to a faulty gene causing liver failure in children. As per statistics from the UMPC Health data, this disease is estimated to affect 1 in 100,000 pediatric patients. With the availability of advanced therapies expanding rapidly, rare disease treatment solutions will gain considerable traction among the pediatric patient population.
Based on the route of administration, rare disease treatment market share from injectable segment is set to depict more than 9% CAGR through 2023-2032, given the high bioavailability of medications administered intravenously. Injectables also provide rapid effects, thereby gaining adoption for the treatment of disorders that need immediate intervention. As a result of the escalating need for accurate and effective therapeutics, the production of injectable orphan drugs will increase, contributing to the rare disease treatment industry dynamics over the forecast period.
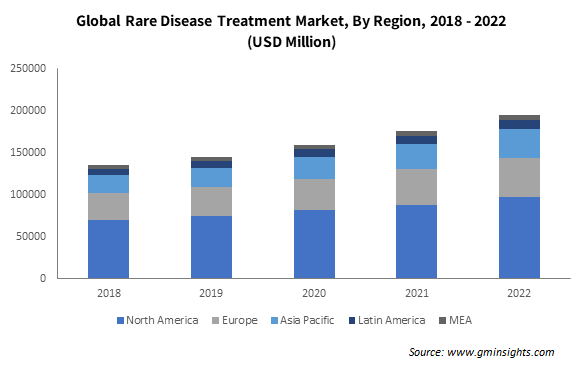
Europe rare disease treatment industry size will exceed USD 115.5 billion by 2032, owing to the rising disease burden and the shifting preference toward early disease diagnosis. Based on data from the European Commission, in 2021, nearly 36 million people were diagnosed with rare diseases in Europe. Such factors are likely to drive up the demand for accurate solutions for early diagnosis, creating strong growth avenues for the rare disease treatment market. Furthermore, the increasing rate of hospitalization and the presence of supportive healthcare policies will proliferate the acceptance of rare disease therapies across the region.

