Summary
Table of Content

U.S. Intrauterine Devices Market
Get a free sample of this report
Form submitted successfully!
Error submitting form. Please try again.
Thank you!
Your inquiry has been received. Our team will reach out to you with the required details via email. To ensure that you don't miss their response, kindly remember to check your spam folder as well!

Request Sectional Data
Thank you!
Your inquiry has been received. Our team will reach out to you with the required details via email. To ensure that you don't miss their response, kindly remember to check your spam folder as well!
Form submitted successfully!
Error submitting form. Please try again.
U.S. Intrauterine Devices Market Size
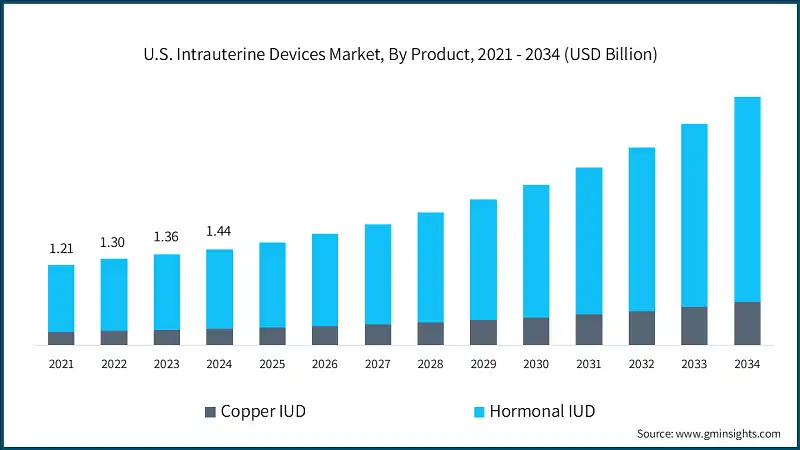
The U.S. intrauterine devices market was valued at USD 1.44 billion in 2024. The market is expected to reach from USD 1.55 billion in 2025 to USD 3.74 billion in 2034, growing at a CAGR of 10.3% from 2025 to 2034, according to the latest report published by Global Market Insights Inc. The high market growth is attributed to the favorable regulatory scenario, rising awareness among women regarding various IUD applications, and a high number of unintended pregnancies, among others.
U.S. Intrauterine Devices Market Key Takeaways
Market Size & Growth
- 2024 Market Size: USD 1.44 Billion
- 2025 Market Size: USD 1.55 Billion
- 2034 Forecast Market Size: USD 3.74 Billion
- CAGR (2025–2034): 10.3%
Regional Dominance
- Largest Market: South Atlantic
- Fastest Growing Country: Mountain States
Key Market Drivers
- Favorable regulatory scenario.
- Rising awareness among women regarding various IUD applications.
- High number of unintended pregnancies.
- Government initiatives for the prevention of unwanted abortions and pregnancies.
- Growing inclination towards planned delayed pregnancy.
- Technological advancements.
Challenges
- High cost of the device.
- Risk of several health issues.
- Variability in insurance coverage and access.
Opportunity
- Rising demand for long-term contraception.
Key Players
- Market Leader: Bayer led with over 49% market share in 2024.
- Leading Players: Top 3 players in this market include AbbVie, Bayer, CooperSurgical, which collectively held a market share of 88% in 2024.
Get Market Insights & Growth Opportunities
Intrauterine devices (IUDs) are essential instruments in contraceptive care, offering women a reversible, highly effective, long-term method of birth control. Key players of the industry are AbbVie, CooperSurgical, and Bayer, among others. These devices are valued for their minimal maintenance, convenience, and ability to provide protection for up to 10 years, depending on the type.
The market has increased from USD 1.21 billion in 2021 and reached USD 1.36 billion in 2023, with a historic growth rate of 8.2%. This growth was primarily driven by rising awareness of contraceptive options, government initiatives promoting family planning, and the increasing adoption of long-acting reversible contraceptives like intrauterine devices.
According to a report published by the United Nations Sexual and Reproductive Health Agency (UNFPA) in 2022, nearly half of all pregnancies, totalling 121 million each year throughout the world, are unintended. These unplanned pregnancies often result in adverse health, social, and economic outcomes, prompting a growing demand for reliable, long-term contraceptive solutions. IUDs, with their over 99% effectiveness and minimal maintenance, are increasingly being adopted as a preferred method of birth control. Their ability to provide extended protection makes them especially appealing to women seeking to avoid unintended pregnancies without the need for daily or monthly intervention.
Intrauterine devices (IUDs) are small, flexible, T-shaped contraceptive tools placed inside the uterus to prevent pregnancy. Designed for long-term use, they offer a reversible and highly effective method of birth control.

To get key market trends
U.S. Intrauterine Devices Market Trends
The U.S. intrauterine devices (IUDs) market has seen several technological advancements and ongoing studies that are enhancing the safety, effectiveness, and user experience of IUDs.
- One of the most significant developments is the use of softer, more flexible materials that improve the patient's experience during insertion and reduce the occurrence of discomfort or complications. Newer IUD designs, such as those with streamlined shapes and more user-friendly insertion mechanisms, have also been introduced, making them more accessible and appealing to healthcare providers and patients alike.
- Additionally, the evolution of hormonal IUDs has made a substantial impact on the contraceptive landscape. These devices release small amounts of hormones locally within the uterus, thereby minimizing systemic side effects. The availability of longer-acting, lower-dose hormonal IUDs has increased their attractiveness to various demographics, particularly among women seeking long-term contraception without the burden of daily pill adherence.
- Moreover, ongoing research and development efforts are focused on creating IUDs that use alternative hormones to levonorgestrel, offering more options for women who may experience sensitivities or adverse reactions to specific hormones. These innovations aim to enhance both the safety and personalization of contraceptive care, ensuring broader accessibility and improved patient outcomes.
U.S. Intrauterine Devices Market Analysis
Learn more about the key segments shaping this market
Based on the product, the U.S. intrauterine devices market is segmented into copper IUD and hormonal IUD. The hormonal IUD segment has asserted its dominance in the market by securing a significant market share of 83% in 2024, owing to its high efficacy, long-term protection, and added benefits such as reduced menstrual bleeding and relief from menstrual cramps. The segment is expected to exceed USD 3.09 billion by 2034, growing at a CAGR of 10.2% during the forecast period.
On the other hand, the copper IUD segment is expected to grow with a CAGR of 10.6%. The growth of this segment can be attributed to the rising demand for non-hormonal contraceptive options, increasing awareness of long-term reversible contraception, and the high efficacy and safety profile of copper IUDs.
- Hormonal IUDs often result in lighter menstrual periods or, in some cases, the complete cessation of menstruation, thereby offering additional health benefits for individuals with heavy or painful periods.
- Moreover, healthcare providers frequently recommend hormonal IUDs for their minimal side effects compared to systemic contraceptive methods, such as the birth control pill. This shift has been instrumental in propelling hormonal IUDs to a dominant market share, with significant percentages of women opting for these devices over other contraceptive options.
Based on age group, the U.S. intrauterine devices market is segmented into 15-19, 20-24, 25-29, 30-34, 35-39, 40-44, and 45+. The 30-34 segment dominated the market in 2024, accounting for USD 366.5 million, and is anticipated to grow at a CAGR of 10.6% during the forecast period.
- The 15-19 segment held a revenue of USD 71.2 million in 2024, with projections indicating a steady expansion at 8.9% CAGR from 2025 to 2034.
- The 20-24 segment was valued at USD 242.8 million in 2024.
- The 25-29 segment held a revenue of USD 327.1 million in 2024, with projections indicating a steady expansion at 10.8% CAGR from 2025 to 2034
- The 35-39 segment is expected to grow with a CAGR of 10.2%
- The 40-44 segment held a market share of 9.9% in 2024.
- The 45+ segment was valued at USD 43.2 million in 2024.
Learn more about the key segments shaping this market
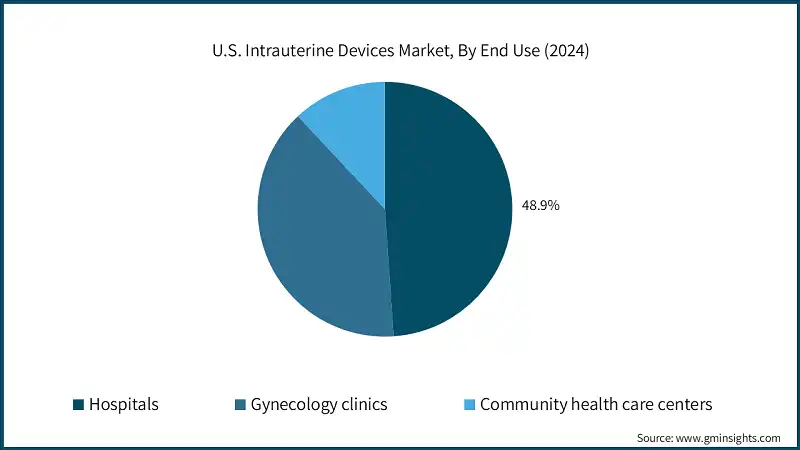
Based on end use, the U.S. intrauterine devices market is classified into hospitals, gynecology clinics, and community health care centers. The hospitals segment dominated the market with a revenue share of 48.9% in 2024 and is expected to reach USD 1.81 billion within the forecast period.
- Hospitals serve as primary access points for long-acting reversible contraception, including IUDs. Their role is reinforced by the presence of trained obstetricians and gynecologists, who ensure safe insertion and removal procedures. Additionally, many hospitals are increasingly providing same-day insertion of IUDs, especially in the postpartum period, which can increase access and decrease unintended pregnancies.
- Additionally, hospitals are central to public health programs targeting underserved populations, often participating in initiatives like the 340B Drug Pricing Program, which subsidizes IUDs for low-income patients. Due to such factors, hospitals are positioned as vital infrastructure for reaching equitable access to IUDs in different demographic groups.
- The gynecology clinics segment held a revenue of USD 564.8 million in 2024, with projections indicating a steady expansion at 10.7% CAGR from 2025 to 2034. This growth is driven by the increasing preference for specialized reproductive care, rising awareness of long-acting contraceptive options, and the growing availability of trained professionals and advanced IUD insertion technologies in outpatient settings.
- The community health care centers segment was valued at USD 173.0 million in 2024, driven by increasing access to affordable reproductive health services and growing adoption of IUDs in underserved and rural populations.
Looking for region specific data?
South Atlantic Intrauterine Devices Market
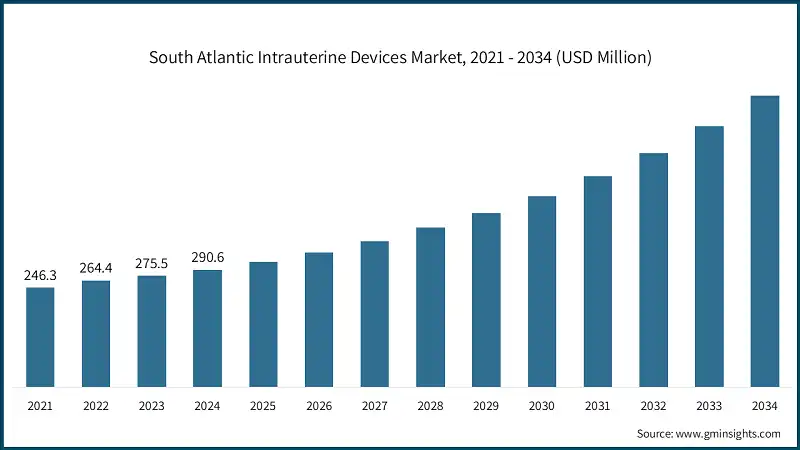
The South Atlantic zone dominated the U.S. intrauterine devices market with the leading market share of 20.1% in 2024.
- This dominance of this region in the U.S. is due to a robust healthcare infrastructure, which includes a wide network of hospitals and gynecology clinic centers that adopt advanced medical technologies for insertion and removal of IUDs. The region also benefits from higher rates of provider training and certification in long-acting reversible contraception (LARC) methods, supported by organizations like the American College of Obstetricians and Gynecologists (ACOG) and state health departments.
- Additionally, the region includes populous states such as Florida, Georgia, and North Carolina, which have high concentrations of reproductive-age women. These systemic advantages, combined with proactive public health outreach and education, contribute to the region’s leadership in IUD adoption and utilization.
Pacific Central Intrauterine Devices Market
The Pacific Central intrauterine devices market accounted for USD 236.8 million in 2024 and is anticipated to show lucrative growth over the forecast period.
- This prominence can be attributed to several factors, including the region’s progressive contraceptive coverage policies, which ensure broader access to IUDs through both public and private insurance plans.
- Additionally, states like California and Washington have implemented provider training programs and same-day insertion protocols, reducing procedural delays and improving adoption of IUDs. Cultural openness toward LARC, combined with strong advocacy from organizations, further supports widespread adoption.
Northeast Intrauterine Devices Market
The Northeast intrauterine devices market is anticipated to grow at the highest CAGR of 10.5% during the analysis timeframe.
- This region is characterized by a diverse population and progressive healthcare policies, leading to an increasing acceptance and utilization of IUDs as a preferred contraceptive method. The combination of urban and rural areas, alongside a higher concentration of healthcare facilities, facilitates awareness and education regarding various contraceptive choices, including IUDs.
- Moreover, states in this region have been proactive in promoting reproductive health services, resulting in enhanced access to contraception and higher adoption rates of IUDs, particularly among younger women and those seeking long-term contraceptive solutions.
U.S. Intrauterine Devices Market Share
Leading industry players such as AbbVie, Bayer, and CooperSurgical hold around 88% of the market share in the competitive market. These companies maintain their leading position by combining strong product lines, business collaborations with healthcare providers, regulatory clearances, and consistent product innovation.
Major companies in the U.S. intrauterine devices market are reinforcing their competitive positions by focusing on innovation, including the development of frameless and biodegradable IUDs, user-friendly insertion systems, and hormone-free alternatives like copper-based spherical designs. Bayer AG continues to lead with its robust hormonal IUD portfolio, including the widely used Mirena and Kyleena systems. The company recently received FDA approval for an extended-duration Mirena IUD, now offering up to 8 years of protection.
Similarly, CooperSurgical is advancing its position through its hormone-free IUD, Paragard, which provides up to 10 years of contraception. The company has launched consumer engagement initiatives and digital tools to improve patient education and access. Its focus on hormone-free solutions appeals to a growing segment of women seeking non-hormonal options.
Meanwhile, other key players, such as AbbVie, DKT, and Sebela Pharmaceuticals, are investing in next-generation IUDs with customizable hormone release, longer duration, and improved biocompatibility. These companies are also exploring biodegradable materials and frameless designs to enhance comfort and reduce side effects.
U.S. Intrauterine Devices Market Companies
A few of the prominent players operating in the U.S. intrauterine devices industry include:
- AbbVie
- Bayer
- CooperSurgical
- DKT
- Medicines360
- Sebela Pharmaceuticals
- AbbVie
AbbVie has made significant strides in democratizing access to hormonal IUDs with Liletta, a cost-effective levonorgestrel-releasing system. Designed for up to 8 years of use, Liletta is FDA-approved and widely adopted in public health clinics across the U.S. AbbVie’s strategy emphasizes affordability, clinical efficacy, and broad accessibility, particularly for underserved populations.
Bayer leads the U.S. IUD market with its flagship hormonal IUDs, which are known for their long duration, high efficacy, and additional therapeutic benefits such as reduced menstrual bleeding. Bayer’s commitment to innovation, sustainability, and expanded access is reflected in its efforts to improve device design and broaden contraceptive availability.
CooperSurgical distinguishes itself with Paragard, an FDA-approved, hormone-free copper IUD in the U.S., offering up to 10 years of protection. The company is investing in consumer engagement platforms and digital education tools to raise awareness about non-hormonal options. CooperSurgical’s focus on reusability, patient comfort, and sustainability aligns with the growing demand for personalized and eco-conscious contraceptive solutions.
U.S. Intrauterine Devices Market Report Attributes
| Key Takeaway | Details |
|---|---|
| Market Size & Growth | |
| Base Year | 2024 |
| Market Size in 2024 | USD 1.44 Billion |
| Market Size in 2025 | USD 1.55 Billion |
| Forecast Period 2025 - 2034 CAGR | 10.3% |
| Market Size in 2034 | USD 3.74 Billion |
| Key Market Trends | |
| Drivers | Impact |
| Favorable regulatory scenario | Streamlined approval processes and supportive policies accelerate market entry and adoption of IUDs. |
| Rising awareness among women regarding various IUD applications | Increased education empowers women to choose long-term, effective contraceptive options. |
| High number of unintended pregnancies | Growing rates of unplanned pregnancies drive demand for reliable birth control methods like IUDs. |
| Government initiatives for the prevention of unwanted abortions and pregnancies | Public health programs promote IUDs as a preventive solution, boosting market growth. |
| Growing inclination towards planned delayed pregnancy | Lifestyle shifts and career planning encourage women to opt for long-term contraception. |
| Technological advancements | Innovations in IUD design and materials enhance comfort, safety, and effectiveness, attracting more users. |
| Pitfalls & Challenges | Impact |
| High cost of the device | Elevated upfront costs can deter adoption, especially among uninsured or low-income populations. |
| Risk of several health issues | Concerns over side effects like cramping, bleeding, or infections may limit patient acceptance. |
| Variability in insurance coverage and access | Inconsistent reimbursement policies create disparities in IUD availability and affordability. |
| Opportunities: | Impact |
| Rising demand for long-term contraception | Growing preference for hassle-free, durable birth control methods is expanding the IUD user base. |
| Market Leaders (2024) | |
| Market Leaders |
49% market share |
| Top Players |
Collective Market Share is 88% |
| Competitive Edge |
|
| Regional Insights | |
| Largest Market | South Atlantic |
| Fastest growing market | Mountain States |
| Emerging states | Arizona, North Dakota, Minnesota, Utah, etc. |
| Future outlook |
|
What are the growth opportunities in this market?
U.S. Intrauterine Devices Industry News:
- In February 2025, Sebela Pharmaceuticals announced that the U.S. Food and Drug Administration (FDA) granted approval of MIUDELLA for the prevention of pregnancy in females of reproductive potential for up to three years. This approval may expand the company’s product offering and help generate enhanced business revenue.
- In November 2023, Bayer and CrossBay Medical Inc. announced a collaboration and licensing agreement to develop and produce a single-handed inserter integrated with Bayer’s leading hormonal intrauterine system (IUS) portfolio. This strategic move has strengthened Bayer’s product offerings and enhanced its competitiveness in the U.S. IUD market by improving ease of use for healthcare providers and comfort for patients.
The U.S. intrauterine devices market research report includes an in-depth coverage of the industry with estimates and forecasts in terms of revenue in USD Million from 2021 - 2034 for the following segments:
Market, By Product
- Copper IUD
- Hormonal IUD
Market, By Age Group
- 15-19
- 20-24
- 25-29
- 30-34
- 35-39
- 40-44
- 45+
Market, By End Use
- Hospitals
- Gynecology clinics
- Community health care centers
The above information is provided for the following zones and states:
- East North Central
- Illinois
- Indiana
- Michigan
- Ohio
- Wisconsin
- West South Central
- Arkansas
- Louisiana
- Oklahoma
- Texas
- South Atlantic
- Delaware
- Florida
- Georgia
- Maryland
- North Carolina
- South Carolina
- Virginia
- West Virginia
- Washington, D.C.
- Northeast
- Connecticut
- Maine
- Massachusetts
- New Hampshire
- Rhode Island
- Vermont
- New Jersey
- New York
- Pennsylvania
- East South Central
- Alabama
- Kentucky
- Mississippi
- Tennessee
- West North Central
- Iowa
- Kansas
- Minnesota
- Missouri
- Nebraska
- North Dakota
- South Dakota
- Pacific Central
- Alaska
- California
- Hawaii
- Oregon
- Washington
- Mountain States
- Arizona
- Colorado
- Utah
- Nevada
- New Mexico
- Idaho
- Montana
- Wyoming
Frequently Asked Question(FAQ) :
What was the market size of the U.S. intrauterine devices in 2024?
The market size was USD 1.44 billion in 2024, driven by a favorable regulatory environment, rising awareness among women about IUD applications, and a high number of unintended pregnancies.
What is the projected value of the U.S. intrauterine devices market by 2034?
The market is expected to reach USD 3.74 billion by 2034, growing at a CAGR of 10.3% from 2025 to 2034.
What is the projected size of the U.S. intrauterine devices market in 2025?
The market is expected to reach USD 1.55 billion in 2025.
How much revenue did the hormonal IUD segment generate in 2024?
The hormonal IUD segment capturing 83% of the market share due to its high efficacy, long-term protection, and additional benefits like reduced menstrual bleeding and relief from menstrual cramps.
What was the valuation of the hospitals segment in 2024?
The hospitals segment held a 48.9% market share in 2024.
Which region leads the U.S. intrauterine devices market?
The South Atlantic zone led the market with a 20.1% share in 2024, driven by a high adoption rate of IUDs and favorable healthcare infrastructure.
What are the upcoming trends in the U.S. intrauterine devices industry?
Key trends include the use of softer, more flexible materials for improved patient comfort, advancements in hormonal IUDs with longer-acting and lower-dose options, and the introduction of streamlined designs for easier insertion.
Who are the key players in the U.S. intrauterine devices market?
Key players include AbbVie, Bayer, CooperSurgical, DKT, Medicines360, and Sebela Pharmaceuticals.
U.S. Intrauterine Devices Market Scope
Related Reports