Hemodynamic Monitoring Devices Market Size & Share 2026-2035
Market Size by Product, by System Type, by End Use, Global Forecast.Report ID: GMI7995
|
Published Date: January 2026
|
Report Format: PDF
Download Free PDF
Authors: Mariam Faizullabhoy, Gauri Wani







Hemodynamic Monitoring Devices Market Size
The global hemodynamic monitoring devices market was valued at USD 1.6 billion in 2025 and is projected to grow from USD 1.7 billion in 2026 to USD 3.1 billion by 2035, expanding at a CAGR of 7.2%, according to the latest report published by Global Market Insights Inc. This substantial growth is driven by numerous factors such as rising incidence of chronic diseases across the globe, technological advancements in hemodynamic monitoring devices, increasing preference for telehealth services, and growing number of surgeries.
Hemodynamic Monitoring Devices Market Key Takeaways
Market Size & Growth
Regional Dominance
Key Market Drivers
Challenges
Opportunity
Key Players
Hemodynamic monitoring devices measure blood pressure and flow within the cardiovascular system in order to evaluate circulation and heart function. They offer real time information on variables like blood pressure, vascular resistance, and cardiac output. These devices are frequently used to direct medication and fluid therapy during high-risk surgeries, critical care units, and operating rooms. Additionally, they are used to treat conditions where accurate cardiovascular monitoring is crucial, such as heart failure, shock, and severe trauma. Koninklijke Philips N.V., Baxter International, Abbott Laboratories, Siemens Healthineers, and GE HealthCare Technologies Inc. are some of the top firms in the hemodynamic monitoring devices market. By introducing cutting-edge non-invasive technologies, incorporating AI-driven analytics, and increasing their presence in emerging markets, leading companies in the market are expanding.
The market grew from USD 1.3 billion in 2022 to USD 1.5 billion in 2024. The rising incidence of chronic diseases worldwide is significantly driving the growth of the market. According to the World Health Organization, noncommunicable diseases (NCDs) caused death of at least 43 million people in 2021, representing 75% of non-pandemic-related deaths globally. Cardiovascular diseases accounted for the largest share with 19 million deaths, followed by cancers (10 million), chronic respiratory diseases (4 million), and diabetes (over 2 million including kidney disease deaths caused by diabetes).
This growing burden of chronic conditions necessitates advanced monitoring solutions to manage hemodynamic parameters effectively, especially in critical care and surgical settings. As these diseases often lead to complications such as heart failure, hypertension, and multi-organ dysfunction, healthcare providers are increasingly adopting hemodynamic monitoring devices to ensure accurate assessment and timely intervention. This adoption is expected to accelerate further with the integration of AI-driven analytics and remote monitoring capabilities, making hemodynamic monitoring an essential component of modern healthcare delivery.
Additionally, the rising volume of surgical procedures is a key factor fueling the growth of the hemodynamic monitoring devices market. These devices are essential for assessing cardiovascular function during complex surgeries, ensuring patient safety and optimizing outcomes. As surgical interventions become more frequent, hospitals and surgical centers are increasingly adopting advanced monitoring systems to manage perioperative risks such as hypotension and cardiac instability.
According to the Agency for Healthcare Research and Quality, U.S. hospital-owned facilities conducted approximately 12.4 million ambulatory surgery encounters in 2022, up from 11.9 million in 2021. This surge in surgical volumes underscores the growing demand for reliable monitoring technologies that can deliver real-time insights into cardiac output, blood pressure, and tissue perfusion. As healthcare systems worldwide expand surgical capacity and prioritize patient safety, the adoption of minimally invasive and advanced hemodynamic monitoring solutions is expected to accelerate, driving market growth over the coming years.
Hemodynamic monitoring devices are a vital part of today's healthcare, providing real-time assessments of cardiovascular parameters including cardiac output, blood pressure, and tissue perfusion. The information recovered from these devices is used to assist clinicians in managing patients who are receiving fluid therapy or undergoing medical treatment during times of surgical or critical care. By using this type of monitoring, clinicians can detect instability in patients' hemodynamic system before severe complications develop, ultimately improving patient outcomes, and providing more individualized treatment plans, making them indispensable in enhancing patient safety and optimizing resource utilization in hospitals and surgical centers.
Hemodynamic Monitoring Devices Market Trends
Hemodynamic Monitoring Devices Market Analysis
Learn more about the key segments shaping this market
Download Free PDF
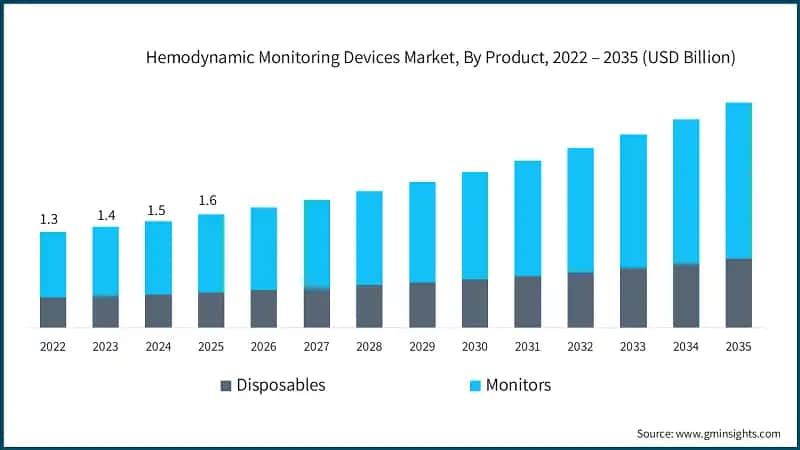
Based on product, the market is bifurcated into disposables and monitors. The monitors segment is dominating the market with the largest revenue of USD 1.1 billion in 2025.
Based on system type, the hemodynamic monitoring devices market is segmented into non-invasive, invasive, and minimally invasive. The non-invasive segment dominated the market with the largest revenue of USD 809.4 million in 2025 and is expected to grow at a CAGR of 7.3% over the forecast period.
Based on end use, the hemodynamic monitoring devices market is segmented into hospitals, ambulatory surgical centers, homecare settings, and other end users. The hospitals segment dominated the market with the largest revenue share of 64.4% in 2025 and is expected to grow at the fastest CAGR over the forecast period.
North America hemodynamic monitoring devices market
North America dominated the market with a market share of 36.4% in 2025.
Europe Hemodynamic Monitoring Devices Market
Europe market accounted for USD 433.9 million in 2025 and is anticipated to show lucrative growth over the forecast period.
Germany hemodynamic monitoring devices market is projected to experience steady growth between 2026 and 2035.
Asia Pacific Hemodynamic Monitoring Devices Market
The Asia Pacific region is projected to be valued at USD 380.7 million in 2025.
Japan hemodynamic monitoring devices market is poised to witness lucrative growth between 2026 - 2035.
Latin America Hemodynamic Monitoring Devices Market
Middle East and Africa Hemodynamic Monitoring Devices Market
The market in Saudi Arabia is expected to experience significant and promising growth from 2026 to 2035.
Hemodynamic Monitoring Devices Market Share
Hemodynamic Monitoring Devices Market Companies
Few of the prominent players operating in the hemodynamic monitoring devices industry include:
Baxter is a major player in the hemodynamic monitoring devices market, notably through its Starling Fluid Management Monitoring System, which utilizes patented bioreactance technology to deliver noninvasive, real-time cardiac output and fluid responsiveness data. This innovation supports individualized fluid therapy across ICU, ED, OR, and rapid response settings, reducing potential complications from invasive lines and aligning with a broader healthcare trend toward non-invasive patient care. Baxter’s strategic focus on non-invasive solutions positions it strongly in a market that increasingly favors safer, patient-friendly monitoring platforms.
Philips holds a significant position in the interventional hemodynamic monitoring market through its Hemo system with IntelliVue X3. Tailored for catheterization labs, this solution integrates advanced hemodynamic measurements into a touchscreen patient monitor, facilitating seamless table side control and real-time data visualization. This approach aligns with Philips’s commitment to workflow optimization and enhanced clinical decision-making in interventional environments, reinforcing its influence in cathlab hemodynamic monitoring.
GE Healthcare’s Mac Lab Hemodynamic Recording System has long been a cornerstone in cath lab monitoring, with nearly 20 years of trusted use. The system excels at integrating imaging, waveform data, and reporting tools within high-volume cath labs, ensuring data synchronization, interoperability, and streamlined clinician workflows. GE’s deep integration with hospital IT systems and its commitment to documentation efficiency underscore its sustained leadership in invasive hemodynamic monitoring platforms.
20% market share
Hemodynamic Monitoring Devices Market News:
The hemodynamic monitoring devices market research report includes an in-depth coverage of the industry with estimates and forecast in terms of revenue in USD Million and from 2022 - 2035 for the following segments:
Click here to Buy Section of this Report
Market, By Product
Market, By System Type
Market, By End Use
The above information is provided for the following regions and countries: