Summary
Table of Content

Real World Evidence Solutions Market
Get a free sample of this report
Form submitted successfully!
Error submitting form. Please try again.
Thank you!
Your inquiry has been received. Our team will reach out to you with the required details via email. To ensure that you don't miss their response, kindly remember to check your spam folder as well!

Request Sectional Data
Thank you!
Your inquiry has been received. Our team will reach out to you with the required details via email. To ensure that you don't miss their response, kindly remember to check your spam folder as well!
Form submitted successfully!
Error submitting form. Please try again.
Real World Evidence Solutions Market Size
The global real world evidence solutions market was valued at USD 2.6 billion in 2025 and is projected to grow from USD 3.1 billion in 2026 to USD 11.9 billion by 2035, expanding at a CAGR of 16.3%, according to the latest report published by Global Market Insights Inc.
Real World Evidence Solutions Market Key Takeaways
Market Size & Growth
- 2025 Market Size: USD 2.6 Billion
- 2026 Market Size: USD 3.1 Billion
- 2035 Forecast Market Size: USD 11.9 Billion
- CAGR (2026–2035): 16.3%
Regional Dominance
- Largest Market: North America
- Fastest Growing Region: Asia Pacific
Key Market Drivers
- Growing focus towards accelerating drug development and cost reduction.
- Growing demand for real-time safety and efficacy monitoring of drugs and medical devices.
- Increasing adoption of RWE solutions for informed reimbursement decision-making.
- Increasing adoption of data analytics services in clinical decision making..
Challenges
- Lack of standardization in integration and interoperability of real-world data.
- Shortage of skilled professionals.
Opportunity
- Emerging therapeutic areas expansion beyond oncology.
- Focus on patient-generated health data integration.
Key Players
- Market Leader: IQVIA Holdings Inc led with over 17% market share in 2025.
- Leading Players: Top 5 players in this market include IQVIA Holdings Inc, IBM Corporation, Flatiron Health, ICON Plc, Fortrea Holdings Corporation, which collectively held a market share of 75% in 2025.
Get Market Insights & Growth Opportunities
This substantial growth is driven by numerous factors such as growing focus towards accelerating drug development and cost reduction, growing demand for real-time safety and efficacy monitoring of drugs and medical devices, increasing adoption of RWE solutions for informed reimbursement decision-making, and increasing adoption of data analytics services in clinical decision making.
Real‑world evidence (RWE) solutions play a key role in modern healthcare by helping organizations analyze data generated during routine clinical practice. Real-world evidence solutions provide the ability to analyze and evaluate clinical, regulatory, and business decisions through the use of health care data and associated information gathered from routine settings such as electronic health records (EHR), claims data, patient registries, and digital health tools. RWE solutions are utilized in many facets of health care, including drug development, clinical trial preparation, regulatory submissions, post-market safety assessment, and value-based care initiatives. The market continues to evolve with advances in cloud computing, artificial intelligence, and data analytics, making it easier to collect, integrate, and interpret large volumes of healthcare data. Major companies operating in the real‑world evidence solutions market include IQVIA Holdings Inc., IBM Corporation, Flatiron Health, ICON plc, and Fortrea Holdings Corporation.
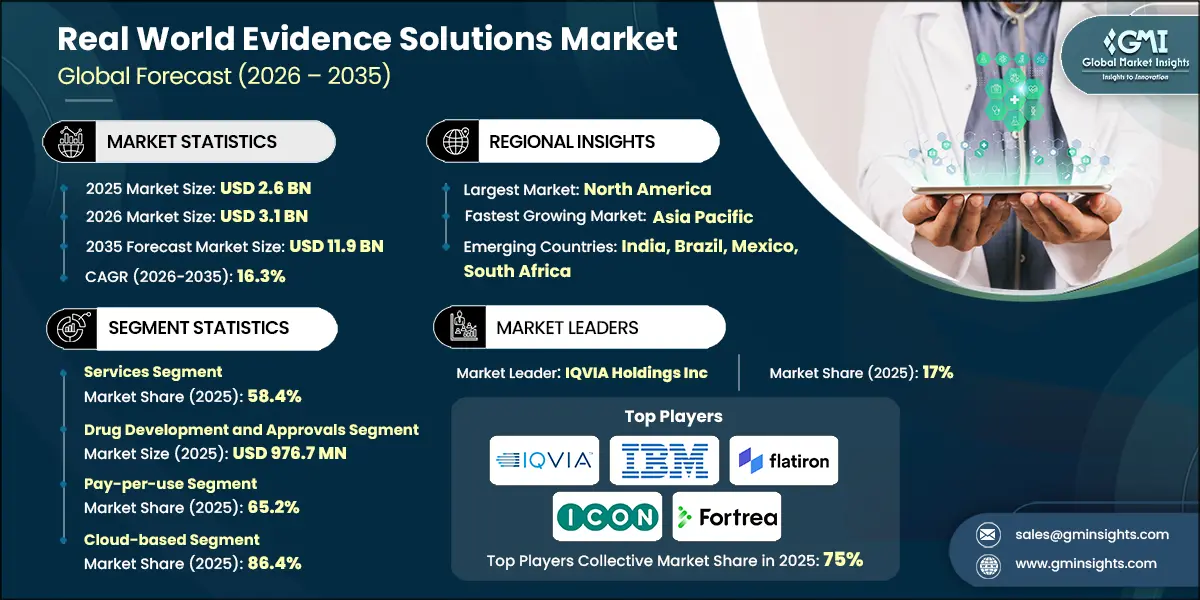
The market grew from USD 1.7 billion in 2022 to USD 2.3 billion in 2024. This growth was driven by the growing focus towards accelerating drug development and cost reduction. Traditional development pathways remain slow and resource‑intensive, relying on lengthy multi‑phase clinical trials and rigorous regulatory evaluations. By leveraging real‑world data from electronic health records, claims databases, and patient registries, RWE supports faster, evidence‑driven decision‑making. It allows biopharmaceutical companies to identify promising drug candidates earlier, assess effectiveness across broader patient groups, and continuously monitor safety in real‑world environments, ultimately helping reduce trial duration, sample size, and overall expenditure. Additionally, RWE is increasingly applied in preclinical phases, where historical clinical datasets complement animal studies by revealing early safety patterns and guiding dose optimization. With regulatory agencies such as the FDA and EMA acknowledging the value of RWE, pharmaceutical firms are accelerating investments in RWE technologies and platforms to facilitate quicker approvals and lower development costs.
Additionally, the shift toward value‑based care is strengthening the need for real‑world evidence solutions in reimbursement decision‑making. Health Technology Assessment (HTA) bodies and payers are increasingly using RWE to assess therapy performance in routine clinical practice, extending evaluation beyond the controlled environment of randomized clinical trials. This supports more transparent, cost‑efficient, and patient‑oriented reimbursement processes. To embed RWE effectively within reimbursement frameworks, Health Technology Assessment (HTA) bodies are developing robust infrastructures that include enhanced analytical capabilities and standardized assessment methodologies. Regulatory organizations are also supporting this transition. For example, the European Medicines Agency has established the DARWIN EU network to deliver timely and reliable RWE for both regulatory and payer evaluations. These efforts enhance consistency, improve trust among stakeholders, and accelerate broader adoption of RWE platforms across global healthcare systems.
Real world evidence solutions encompass a wide variety of methods, technologies, and services used to generate insight into aspects of health care. RWE solutions utilise data collected outside of the clinical trial environment, as well as other sources such as registries, observational studies to better understand health care outcomes, treatment effectiveness, and patients’ experience.
To get key market trends
Real World Evidence Solutions Market Trends
- The growing demand for real‑time safety and efficacy monitoring of drugs and medical devices is accelerating the adoption of advanced real‑world evidence (RWE) solutions. Healthcare stakeholders increasingly require continuous, data‑driven insights into product performance across diverse patient populations to support timely risk identification and treatment effectiveness evaluation.
- Traditional post-marketing surveillance methods, which rely on data that is collected later or sporadically, are not able to meet these requirements. As a result, the market is beginning to move towards RWE platforms that can provide real time insights so that proper safety measures are taken and clinical decision-making is informed.
- Flatiron Health exemplifies this trend through its oncology‑focused RWE offerings built on continuously updated clinical data captured via the OncoEMR system. By enabling life‑science companies to analyze treatment patterns, safety outcomes, and real‑world effectiveness at scale, Flatiron supports faster and more informed evidence‑based decisions.
- As real‑time safety and efficacy monitoring becomes both an operational and regulatory necessity, RWE platforms are evolving from optional analytical tools into critical infrastructure. This sustained demand for continuous, scalable real‑world insights is expected to support long‑term growth of the RWE solutions market.
Real World Evidence Solutions Market Analysis
Learn more about the key segments shaping this market
Based on the component, the real-world evidence solutions market is segmented into services and data sets The services segment held a significant market share of 58.4% in 2025.
- The services segment is a core component of the global real‑world evidence (RWE) solutions market, providing essential support for the design, execution, and regulatory acceptance of RWE studies. These services enable healthcare and life‑science organizations to generate reliable real‑world insights across diverse healthcare systems and regulatory environments.
- Data collection and management services play a critical role by enabling the integration of multiple data sources, including electronic health records, claims databases, registries, wearable devices, and patient‑reported outcomes. Service providers ensure data quality, standardization, and regulatory compliance across regions. For example, Syneos Health offers source‑agnostic data collection services through its Real World & Late Phase Research (RWLP) division, integrating data from clinical sites, registries, claims, and wearable technologies using advanced analytics and AI/ML capabilities.
- In addition, cloud‑based platforms and AI‑driven data harmonization tools are increasingly used to automate and scale RWE processes globally. These technologies support efficient integration of structured and unstructured data while enabling advanced analytics, such as predictive modeling, comparative effectiveness research, and regulatory reporting.
- As healthcare systems worldwide continue to adopt data‑driven decision‑making, the services segment is playing a vital role in expanding global RWE adoption. By addressing data complexity and scalability challenges, service providers are supporting the broader use of RWE across global markets.
Based on application, the real world evidence solutions market is segmented into drug development and approvals, medical device development and approvals, post-market surveillance, market access and reimbursement/coverage decision-making and clinical and regulatory decision-making. The drug development and approvals segment was worth USD 976.7 million in 2025.
- The drug development and approvals segment holds a leading position in the real‑world evidence (RWE) market due to the increasing complexity, cost, and risk of traditional drug development processes. Pharmaceutical companies are increasingly adopting RWE to support clinical trial planning, optimize study design, and improve decision‑making using real‑world data sources such as electronic health records, claims databases, and registries.
- RWE is widely used to identify suitable patient populations, enhance trial efficiency, and monitor long‑term safety and treatment effectiveness beyond controlled trial settings. By complementing clinical trial data with real‑world insights, drug developers can better understand how therapies perform in routine clinical practice and reduce development timelines.
- Regulatory authorities, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), are actively encouraging the use of RWE in regulatory decision‑making. Initiatives such as the FDA’s RWE Framework support its application in label expansions, post‑approval studies, and new indication approvals, prompting pharmaceutical companies to integrate RWE earlier in the development lifecycle.
- The use of RWE is particularly valuable in therapeutic areas such as rare diseases and oncology, where traditional randomized controlled trials are often limited by small patient populations. In these cases, RWE supports accelerated approval pathways, AI‑enabled analytics, and real‑time monitoring, thus helping drive overall market growth.
Based on revenue model, the real world evidence solutions market is segmented into pay-per-use (value-based pricing) and subscription model. The pay-per-use segment held a significant market share of 65.2% in 2025.
- The pay‑per‑use revenue model is an important segment in the real‑world evidence (RWE) market, offering flexible, on‑demand access to data and analytics services. Under this model, pharmaceutical companies, contract research organizations, and healthcare providers can purchase specific RWE services without entering long‑term contracts or subscribing to full platforms.
- This approach typically includes access to curated patient datasets, disease registries, claims databases, and AI‑enabled analytics for targeted research or regulatory activities. By allowing users to select only the required datasets or analytical capabilities, the pay‑per‑use model reduces upfront costs and improves cost efficiency.
- The model enables organizations to scale their RWE activities based on individual project needs, making it well suited for early‑stage drug development, post‑market surveillance, and health economics and outcomes research. This flexibility supports faster decision‑making and encourages adoption among smaller companies and early‑stage programs.
- Several RWE providers, including Aetion and TriNetX, support pay‑per‑use or flexible access models that allow users to obtain services such as patient cohort identification, protocol feasibility assessment, or post‑market safety analysis as needed. By improving affordability and scalability, this revenue model is supporting wider adoption of RWE solutions and contributing to overall market growth.
Based on deployment model, the real world evidence solutions market is segmented into on-premises and cloud-based model. The cloud-based segment held a significant market share of 86.4% in 2025.
- Cloud‑based deployment refers to delivering real‑world evidence (RWE) solutions through remote servers hosted on the internet, enabling users to access data storage, analytics tools, and computing resources on‑demand without the need for local infrastructure.
- The cloud‑based segment plays a critical role in the growth of the RWE market by efficiently managing the increasing volume and complexity of real‑world data from sources such as EHRs, claims, registries, and wearable devices. Scalable computing power and flexible storage support faster data integration and analysis. For example, Optum (UnitedHealth Group) offers cloud‑based RWE and HEOR platforms that leverage claims data, EHRs, and longitudinal patient records to support large‑scale evidence generation.
- In addition, the rising use of artificial intelligence and machine learning in RWE analytics has accelerated the adoption of cloud deployment. Advanced applications such as predictive modeling, cohort identification, and real‑time insights require high‑performance and elastic computing environments, which cloud platforms are well equipped to deliver, enabling faster and more accurate decision‑making.
- Cloud‑based deployment also improves collaboration among pharmaceutical companies, CROs, and regulators by enabling secure and shared access to data environments. Strong security frameworks and compliance with regulations such as HIPAA and GDPR further increase confidence in cloud adoption for sensitive healthcare data, thus helping market growth.
Learn more about the key segments shaping this market
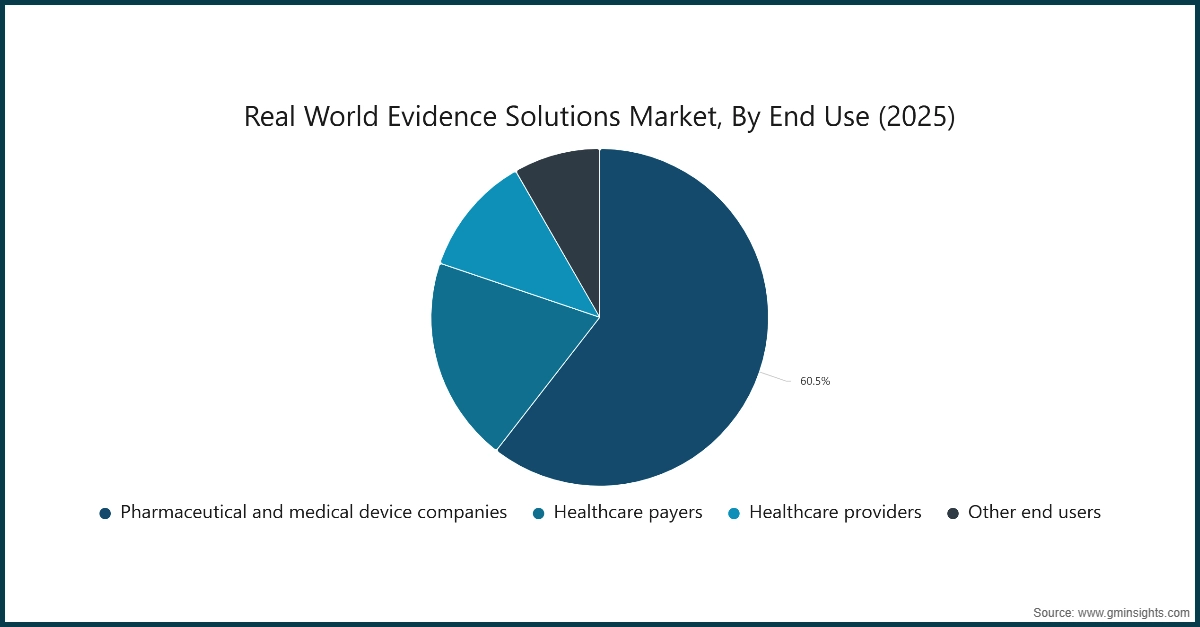
Based on end use, the real world evidence solutions market is categorized into pharmaceutical and medical device companies, healthcare payers, healthcare providers and other end users. Among these, the pharmaceutical and medical device companies segment dominated the market in 2025, accounting for 60.5% of the total market share. This dominance is projected to continue, with the segment expected to reach USD 7.4 billion by 2035.
- Pharmaceutical and medical device companies are among the primary end users of real‑world evidence (RWE) solutions. They apply RWE across the full product lifecycle, including early‑stage drug discovery, clinical trial design, regulatory submissions, post‑market surveillance, and market access planning. This broad adoption reflects the growing need for real‑world insights beyond controlled clinical environments.
- RWE supports the optimization of clinical trial design by identifying suitable patient populations, improving protocol feasibility, and reducing development timelines. It also enables the generation of regulatory‑grade evidence that complements or, in certain cases, substitutes traditional randomized controlled trials, especially in rare diseases and post‑approval studies.
- The increasing acceptance of RWE by regulatory authorities such as the U.S. FDA has further accelerated adoption among pharmaceutical and device manufacturers. As a result, these companies are investing significantly in RWE platforms, analytics capabilities, and data partnerships to strengthen regulatory submissions and support label expansions.
- Overall, driven by strong innovation pipelines, strict regulatory requirements, and the need to demonstrate real‑world value for market access, pharmaceutical and medical device companies play a pivotal role in expanding the global RWE solutions market.
Looking for region specific data?
North America Real World Evidence Solutions Market
North America dominated the global market with a market share of 43.3% in 2025.
- North America dominates the real‑world evidence (RWE) solutions market, driven by the high prevalence of chronic diseases such as cardiovascular conditions and cancer, which require continuous monitoring and data‑driven care interventions. The region benefits from advanced healthcare infrastructure, widespread adoption of electronic health records, and strong penetration of cloud‑based and AI‑enabled analytics platforms. These capabilities allow healthcare providers, payers, and life sciences companies to generate timely insights, identify high‑risk populations, support preventive care, and monitor real‑world treatment effectiveness.
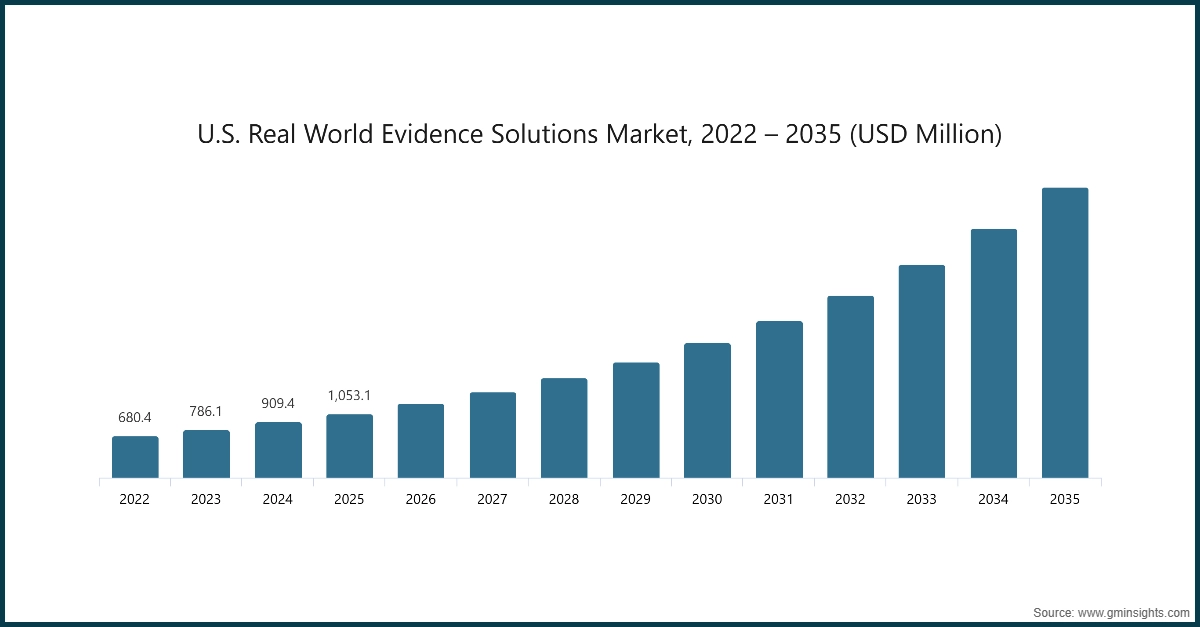
- The U.S. real world evidence solutions market was valued at USD 680.4 million and USD 786.1 million in 2022 and 2023, respectively. The market size reached USD 1,053.1 million in 2025, growing from USD 909.4 million in 2024.
- Cancer represents a major public health challenge in North America and is a key factor accelerating RWE adoption. In the U.S. alone, an estimated 2,041,910 new cancer cases are expected in 2025, with 618,120 deaths projected. The cancer incidence rate stands at 445.8 per 100,000, while the mortality rate is 145.4 per 100,000. As of January 2022, there were 18.1 million cancer survivors, a figure projected to reach 26 million by 2040. These trends highlight the growing need for longitudinal data analysis, post‑treatment monitoring, and survivorship care planning, areas where RWE platforms play a critical role in tracking outcomes and optimizing therapies. Supported by favorable regulatory frameworks and strong acceptance of RWE by authorities such as the FDA, North America continues to lead in RWE adoption and innovation, thus helping market growth.
Europe Real World Evidence Solutions Market
Europe market accounted for USD 719.6 million in 2025 and is anticipated to show lucrative growth over the forecast period.
- Europe is witnessing steady growth in the real‑world evidence (RWE) market, supported by a high prevalence of chronic diseases and a well‑established public healthcare system. The region’s strong emphasis on data‑driven healthcare delivery and evidence‑based policymaking has positioned RWE as a critical tool for improving clinical outcomes and informing regulatory and reimbursement decisions across healthcare systems.
- According to the World Health Organization (WHO), Europe records more than 3.7 million new cancer cases and approximately 1.9 million cancer‑related deaths annually. In 2021, cancer accounted for 21.6% of all deaths in the European Union, underscoring the significant disease burden and the growing need for real‑world data to support early diagnosis, treatment evaluation, and population‑level health planning.
- Cancer continues to represent a major public health challenge across the region, driving increased adoption of RWE platforms. These solutions are increasingly used to support early detection, optimize treatment pathways, and monitor long‑term patient outcomes, particularly in oncology where personalized medicine is rapidly advancing. By enabling analysis of diverse and longitudinal patient data, RWE tools help healthcare stakeholders identify care gaps and improve the effectiveness and efficiency of cancer care delivery.
Germany real world evidence solutions market is projected to experience steady growth between 2026 and 2035.
- Germany is an emerging market for real‑world evidence (RWE) solutions in Europe, driven by a high burden of chronic and complex diseases along with a well‑developed healthcare infrastructure. The country’s strong hospital networks, research institutions, and structured health insurance system generate extensive real‑world data, creating favorable conditions for RWE adoption across clinical and policy settings.
- The growing use of RWE in Germany is supported by a strong commitment to evidence‑based healthcare, increasing demand for personalized medicine, and the need to improve clinical and reimbursement decision‑making. RWE platforms are increasingly being integrated into national cancer strategies and clinical guidelines to enable more targeted, efficient, and outcome‑driven care delivery, thereby supporting continued market growth.
Asia Pacific Real World Evidence Solutions Market
The Asia Pacific region is projected to be valued at USD 548.5 million in 2025.
- The Asia Pacific region is witnessing rapid growth in the adoption of real‑world evidence (RWE) solutions, driven by rising healthcare demands, demographic transitions, and increasing emphasis on data‑driven decision‑making. The region faces a growing burden of chronic and age‑related diseases such as cancer, cardiovascular disorders, creating strong demand for scalable and accurate real‑world data analytics across diverse healthcare systems.
- According to WHO, cancer remains a major public health concern in Asia Pacific, with the region reporting nearly 9.8 million new cases and 5.46 million deaths annually. These figures highlight the urgent need for real‑world data to support early detection, evaluate treatment effectiveness, and improve long‑term survivorship care. RWE platforms enable stakeholders to analyze large and heterogeneous patient populations, supporting more informed clinical and policy decisions.
- Additionally, increasing digital health adoption, expanding healthcare infrastructure, and evolving regulatory frameworks are accelerating RWE uptake across the region. Governments, healthcare providers, and life sciences companies are increasingly leveraging RWE to improve clinical outcomes, align with regulatory requirements, and optimize healthcare resource planning, thereby supporting sustained market growth in Asia Pacific.
Japan real world evidence solutions market is poised to witness lucrative growth between 2026 – 2035.
- Japan’s real‑world evidence (RWE) market is increasingly shaped by the growing demand for real‑time safety and efficacy monitoring of drugs and medical devices, particularly in the post‑approval phase. With a highly regulated healthcare environment and a rapidly aging population, Japan places strong emphasis on continuous evaluation of therapeutic performance under routine clinical practice. RWE platforms enable timely detection of adverse events, assessment of treatment effectiveness across broader patient populations, and ongoing benefit‑risk evaluation, supporting safer and more responsive healthcare delivery.
- As a result, pharmaceutical and device manufacturers are increasingly investing in RWE solutions to meet regulatory expectations, improve post‑market compliance, and generate timely evidence, thereby driving sustained growth of the RWE market in Japan.
Latin America Real World Evidence Solutions Market
- Brazil is gradually expanding its real‑world evidence (RWE) market, supported by a large and diverse patient population, a rising burden of chronic diseases, and increasing digitization of healthcare systems. The country’s public health system generates extensive real‑world data from hospitals, primary care networks, and disease registries, creating growing opportunities for RWE applications in population health management and treatment evaluation.
- Adoption of RWE in Brazil is further driven by the need to support regulatory assessments, improve access decisions, and evaluate the real‑world performance of therapies in a cost‑constrained environment. Pharmaceutical and medical device companies are increasingly using RWE to demonstrate value, address regional treatment variability, and support health technology assessment discussions, while government initiatives focused on data integration are improving long‑term analytical capabilities.
Middle East and Africa Real World Evidence Solutions Market
The real-world evidence solutions market in Saudi Arabia is expected to experience significant and promising growth from 2026 to 2035.
- Saudi Arabia is emerging as a promising market for RWE solutions, driven by healthcare system modernization under Vision 2030 and rising prevalence of chronic and lifestyle‑related diseases such as diabetes, cardiovascular disorders, and cancer. Significant investments in digital health infrastructure, national registries, and electronic health records are strengthening the foundation for real‑world data collection and analysis.
- The growing use of RWE in Saudi Arabia is also supported by regulatory and payer interest in outcome‑based healthcare models and post‑market monitoring of drugs and medical devices. RWE platforms are increasingly being used to inform clinical guidelines, assess treatment effectiveness in local populations, and support value‑based care initiatives, positioning the country as a regional hub for data‑driven healthcare innovation.
Real World Evidence Solutions Market Share
- Leading companies such as IQVIA Holdings Inc., Fortrea Holdings Inc, IBM Corporation, Flatiron Health, and ICON plc are steadily expanding their global presence through a combination of strategic mergers, partnerships, and innovation‑focused initiatives. Together, these market leaders account for an estimated 75% share of the global RWE solutions market, reflecting their strong influence and broad customer reach.
- A key strategic priority for these players is the integration of artificial intelligence and advanced analytics into RWE platforms. By embedding AI‑driven tools into clinical and research workflows and supporting decentralized and hybrid trial models, they are improving the speed, accuracy, and scalability of real‑world evidence generation across diverse therapeutic areas.
- In parallel, collaborations with regulatory authorities and healthcare stakeholders are central to their growth strategies. Close engagement with regulators helps ensure that RWE solutions remain aligned with evolving compliance and evidence standards, while efforts to expand data access to underserved and diverse patient populations are increasing the inclusiveness and real‑world applicability of evidence generation.
- Overall, through continuous data‑driven innovation, these leading RWE providers are enhancing healthcare intelligence and supporting better patient outcomes. Their growing focus on real‑world data is reshaping clinical research, regulatory decision‑making, and population‑level health planning across global healthcare systems.
Real World Evidence Solutions Market Companies
Few of the prominent players operating in the real world evidence solutions industry include:
- Aetion, Inc.
- Cytel Inc
- Flatiron Health Inc
- Fortrea Holdings Inc
- IBM Corporation
- ICON plc
- IQVIA Holdings Inc.
- Medidata Solutions, Inc.
- Merative
- Oracle Corporation
- Parexel International Corporation
- Syneos Health Inc
- Tempus
- TriNetX
- Thermo Fisher Scientific, Inc
- UnitedHealth Group Incorporated
IQVIA Holdings Inc. is a global leader in the real‑world evidence solutions market, offering comprehensive RWE platforms that support the entire life‑sciences value chain. The company leverages large‑scale real‑world data from electronic health records, claims, registries, and other sources, combined with advanced analytics and AI, to support clinical development, regulatory submissions, health economics and outcomes research (HEOR), and post‑market surveillance.
IBM Corporation plays an important role in the RWE solutions market through its cloud‑based healthcare analytics and artificial intelligence capabilities. Through platforms such as IBM Watson for Real‑World Evidence, the company enables life sciences and healthcare organizations to integrate and analyze complex real‑world datasets, supporting evidence generation, safety monitoring, and research decision‑making at scale.
Flatiron Health is a leading provider of oncology‑focused real‑world evidence solutions, specializing in high‑quality, curated data derived from electronic health records. Its RWE platforms support oncology drug development, regulatory interactions, comparative effectiveness research, and market access strategies by generating real‑world insights across the cancer care continuum.
Real World Evidence Solutions Market Report Attributes
| Key Takeaway | Details |
|---|---|
| Market Size & Growth | |
| Base Year | 2025 |
| Market Size in 2025 | USD 2.6 Billion |
| Market Size in 2026 | USD 3.1 Billion |
| Forecast Period 2026-2035 CAGR | 16.3% |
| Market Size in 2035 | USD 11.9 Billion |
| Key Market Trends | |
| Drivers | Impact |
| Growing focus towards accelerating drug development and cost reduction | Drives demand for efficient RWE platforms to streamline trials and reduce research and development expenses. |
| Growing demand for real-time safety and efficacy monitoring of drugs and medical devices | Boosts adoption of RWE tools for post-market surveillance and regulatory compliance. |
| Increasing adoption of RWE solutions for informed reimbursement decision-making | Encourages payers and providers to rely on evidence-based value assessments, expanding market scope. |
| Increasing adoption of data analytics services in clinical decision making. | Enhances the utility of RWE in personalized medicine and treatment optimization. |
| Pitfalls & Challenges | Impact |
| Lack of standardization in integration and interoperability of real-world data | Limits scalability and consistency of RWE insights across platforms and stakeholders. |
| Shortage of skilled professionals | Slows adoption and innovation due to gaps in data science, regulatory, and clinical expertise. |
| Opportunities: | Impact |
| Emerging therapeutic areas expansion beyond oncology | Broadens the application of RWE across neurology, cardiology, and rare diseases, unlocking new market segments. |
| Focus on patient-generated health data integration | Enhances data richness and personalization, driving innovation in treatment and care delivery models. |
| Market Leaders (2025) | |
| Market Leader |
17% Market Share |
| Top Players |
Collective market share is 75% in 2025 |
| Competitive Edge |
|
| Regional Insights | |
| Largest Market | North America |
| Fastest growing market | Asia Pacific |
| Emerging countries | India, Brazil, Mexico, South Africa |
| Future outlook |
|
What are the growth opportunities in this market?
Real World Evidence Solutions Industry News
- In February 2021, Parexel and NeoGenomics announced a strategic collaboration to enhance precision medicine in oncology trials. The partnership aimed to integrate real-world genomics data to improve patient matching, optimize trial design, and support translational research. By linking genomic insights with clinical datasets, the collaboration enabled more targeted recruitment and faster enrollment of patients with specific cancer mutations.
- In March 2020, Syneos Health partnered with ConcertAI to enhance oncology clinical trials by integrating real-world evidence (RWE), focusing on external control arms and regulatory-grade data to accelerate drug development, especially for rare and complex cancers.
- In April 2023, TriNetX announced a strategic partnership with Norstella, acquiring its subsidiary Clinerion Ltd. to expand its global real-world data (RWD) research network. This acquisition boosts TriNetX’s reach and analytical depth in the RWE, supporting faster, more informed drug development.
The real-world evidence solutions market research report includes an in-depth coverage of the industry with estimates and forecast in terms of revenue in USD Million and from 2022 – 2035 for the following segments:
Market, By Component
- Services
- Data collection and integration
- Study design and execution
- Prospective observational studies
- Retrospective database studies
- Site-centric studies
- Registry-based studies
- Hybrid studies
- Regulatory and market access support
- Evidence network
- Other services
- Data sets
- Disparate data sets
- Clinical settings data sets
- Claims data sets
- Pharmacy data sets
- Patient powered data sets
- Registry-based data sets
- Integrated data sets
- Disparate data sets
Market, By Application
- Drug development and approvals
- Oncology
- Cardiovascular disease
- Neurology
- Immunology
- Other therapeutic areas
- Medical device development and approvals
- Post-market surveillance
- Market access and reimbursement/coverage decision-making
- Clinical and regulatory decision-making
Market, By Revenue Model
- Pay-per-use (value-based pricing)
- Subscription
Market, By Deployment Model
- On-premise
- Cloud-based
Market, By End Use
- Pharmaceutical and medical device companies
- Healthcare payers
- Healthcare providers
- Other end users
The above information is provided for the following regions and countries:
- North America
- U.S.
- Canada
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Netherlands
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- Latin America
- Brazil
- Mexico
- Argentina
- Middle East and Africa
- Saudi Arabia
- South Africa
- UAE
Frequently Asked Question(FAQ) :
What is the real-world evidence solutions market size in 2025?
The market size for real-world evidence solutions is valued at USD 2.6 billion in 2025. The increasing focus on accelerating drug development and reducing costs drives the demand for efficient RWE platforms.
What is the market size of the real-world evidence solutions industry in 2026?
The market size for real-world evidence solutions reached USD 3.1 billion in 2026, reflecting steady growth driven by the adoption of RWE tools for regulatory compliance and post-market surveillance.
What is the projected value of the real-world evidence solutions market by 2035?
The market size for real-world evidence solutions is expected to reach USD 11.9 billion by 2035, growing at a CAGR of 16.3%. This growth is fueled by the integration of patient-generated health data, expansion into new therapeutic areas, and advancements in data analytics.
How much revenue did the services segment generate in 2025?
The services segment accounted for 58.4% of the market in 2025, making it the largest component category. Its dominance is supported by the growing demand for data analytics services and consulting solutions in clinical decision-making.
What was the valuation of the drug development and approvals segment in 2025?
The drug development and approvals segment was valued at USD 976.7 million in 2025, driven by the increasing reliance on RWE for streamlining clinical trials and expediting regulatory approvals.
What is the growth outlook for the pharmaceutical and medical device companies segment from 2025 to 2035?
The pharmaceutical and medical device companies segment is projected to grow significantly, reaching USD 7.4 billion by 2035. This growth is attributed to the rising adoption of RWE solutions for drug and device development, as well as post-market surveillance.
Which region leads the real-world evidence solutions market?
North America dominated the market in 2025, holding a 43.3% share. The region's leadership is driven by advancements in healthcare IT infrastructure, increased adoption of RWE tools, and strong regulatory support for evidence-based decision-making.
What are the upcoming trends in the real-world evidence solutions industry?
Key trends include the integration of patient-generated health data, expansion into therapeutic areas beyond oncology, adoption of cloud-based deployment models, and advancements in AI-driven data analytics.
Who are the key players in the real-world evidence solutions market?
Key players include IQVIA Holdings Inc., IBM Corporation, Flatiron Health, ICON Plc, Fortrea Holdings Corporation, Aetion, Inc., Cytel Inc., Medidata Solutions, Inc., Merative, Oracle Corporation, and Parexel International Corporation.
Real World Evidence Solutions Market Scope
Related Reports