Summary
Table of Content

Pharmaceutical Stability and Storage Services Market
Get a free sample of this report
Form submitted successfully!
Error submitting form. Please try again.
Thank you!
Your inquiry has been received. Our team will reach out to you with the required details via email. To ensure that you don't miss their response, kindly remember to check your spam folder as well!

Request Sectional Data
Thank you!
Your inquiry has been received. Our team will reach out to you with the required details via email. To ensure that you don't miss their response, kindly remember to check your spam folder as well!
Form submitted successfully!
Error submitting form. Please try again.
Pharmaceutical Stability and Storage Services Market Size
The growing focus on regulatory compliance, increasing investments in drug development and research, and expansion in global supply chains are collectively driving the revenue growth in the market.
The growing focus on regulatory compliance is significantly driving the growth of the market, as stringent global regulations mandate precise stability and storage protocols to ensure drug safety and efficacy. Agencies such as the U.S. Food and Drug Administration (U.S. FDA) and the European Medicines Agency (EMA) require detailed stability testing data and compliance with guidelines such as International Council for Harmonisation (ICH) Q1A (R2), compelling pharmaceutical companies to invest in advanced stability studies and storage facilities.
Pharmaceutical Stability and Storage Services Market Key Takeaways
Market Size & Growth
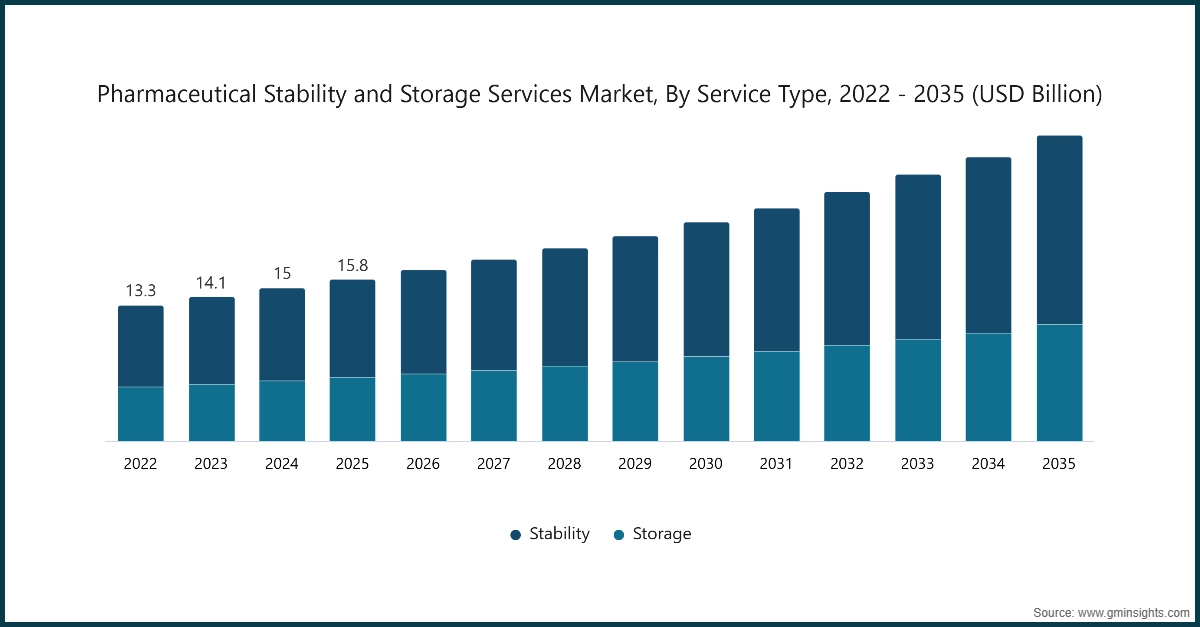
- 2025 Market Size: USD 15.8 Billion
- 2026 Market Size: USD 16.8 Billion
- 2035 Forecast Market Size: USD 29.9 Billion
- CAGR (2026–2035): 6.6%
Regional Dominance
- Largest Market: North America
- Fastest Growing Region: Asia Pacific
Key Market Drivers
- Growing focus on regulatory compliance.
- Increasing investments in drug development and research.
- Technological innovations enhancing pharmaceutical stability and storage.
- Expansion in global supply chains.
Challenges
- High cost associated with specialized storage solutions.
- Concerns related to transportation and logistics.
Opportunity
- Rising demand for end‑to‑end outsourced stability and storage ecosystems.
Key Players
- Market Leader: Catalent led with over 15% market share in 2025.
- Leading Players: Top 5 players in this market include Catalent, Charles River Laboratories, Eurofins Scientific, Intertek Group, Almac Group, which collectively held a market share of 45% in 2025.
Get Market Insights & Growth Opportunities
Additionally, in 2023, the U.S. FDA reported that the top three reasons for drug recalls were related to manufacturing quality issues, including contamination and improper storage conditions, further highlighting the need for adherence to rigorous standards, driving market growth. This focus ensures that pharmaceuticals maintain quality throughout their lifecycle, fostering demand for specialized stability and storage services. Moreover, the top 5 players in this market are Catalent, Charles River Laboratories, Eurofins Scientific, Intertek Group, and Almac Group.
Pharmaceutical stability and storage services refer to specialized solutions provided to ensure that pharmaceutical products, including drugs, biologics, and medical devices, maintain their intended efficacy, safety, and quality throughout their shelf life. These services involve controlled storage under specified conditions (e.g., temperature, humidity, and light) and systematic stability testing to evaluate the product's behavior over time.
The global pharmaceutical stability and storage services market was estimated at USD 15.8 billion in 2025. The market is expected to grow from USD 16.8 billion in 2026 to USD 29.9 billion by 2035 at a CAGR of 6.6%, according to the latest report published by Global Market Insights Inc.
To get key market trends
Pharmaceutical Stability and Storage Services Market Trends
- Technological innovations are revolutionizing the pharmaceutical stability and storage domain, ensuring improved accuracy, compliance, and efficiency. The integration of advanced monitoring systems, such as Internet of Things (IoT)-enabled devices, allows for real-time temperature and humidity tracking, which is critical for maintaining the stability of sensitive pharmaceutical products.
- Additionally, developments in cryogenic storage technologies, such as liquid nitrogen-based systems, are facilitating the safe storage of cellular therapies and mRNA-based vaccines. Automated robotic storage systems are also gaining traction, offering better inventory management and minimizing human error.
- Technological innovations are redefining the standards for pharmaceutical stability and storage by ensuring enhanced precision and efficiency in maintaining optimal storage conditions. Advancements in environmental monitoring systems have significantly improved the ability to track critical parameters such as temperature, humidity, and light exposure. These systems not only help in meeting regulatory requirements but also minimize the risk of product degradation, ensuring the safety and efficacy of pharmaceutical products. Such innovations are particularly crucial for biologics and temperature-sensitive drugs, where even minor fluctuations in storage conditions can impact product stability.
- These innovations are driving the pharmaceutical stability and storage services market by providing cost-effective, reliable, and compliant solutions, catering to the increasing demand for precise storage of high-value drugs. The adoption of these technologies is helping manufacturers meet stringent regulatory requirements, thus boosting the market's growth.
Pharmaceutical Stability and Storage Services Market Analysis
Learn more about the key segments shaping this market
Based on service type, the pharmaceutical stability and storage services market is segmented into stability and storage. Stability segment is further sub-segmented into drug substance, stability indicating method validation, accelerated stability testing, photostability testing, and other stability testing methods. Similarly, the storage segment is further sub-segmented into cold and non-cold. The stability segment dominates the market with a share of 60.8% in 2025.
- Stability testing services are essential for ensuring the safety and effectiveness of drugs throughout their lifecycle. Regulatory agencies such as the U.S. FDA and EMA require stability testing for drug substances and finished products, which increases the demand for these services.
- These tests check how factors such as temperature, humidity, and light affect drugs, ensuring compliance with Good Manufacturing Practices (GMP) and regulatory requirements.
- Additionally, the growing complexity of drug formulations, such as biologics and biosimilars, requires advanced stability testing methods. Techniques such as accelerated stability testing and photostability analysis help determine a drug's shelf life and storage conditions. As the pharmaceutical industry focuses on creating innovative drugs, stability testing remains a critical part of the market.
Based on molecule type, the pharmaceutical stability and storage services market is segmented into small molecule and large molecule. The small molecule and large molecule are each sub-segmented into commercial products and research products. The small molecule segment dominated the market with a share of 54.7% in 2025.
- Small molecules are extensively used in drug development and commercialization. These molecules form the backbone of most oral medications, which constitute the majority of pharmaceutical products globally.
- The established manufacturing processes, cost-effectiveness, and scalability of small molecule drugs make them widely preferred, driving consistent demand for stability and storage services tailored to their specific requirements.
- Furthermore, the global market for small molecule drugs is well-established, with a steady pipeline of generic and innovative drugs entering the market.
- Regulatory authorities mandate rigorous stability testing for small molecules to ensure their efficacy and safety across diverse environmental conditions. This sustained demand for compliance further strengthens the dominance of the small molecule segment in the market.
Learn more about the key segments shaping this market
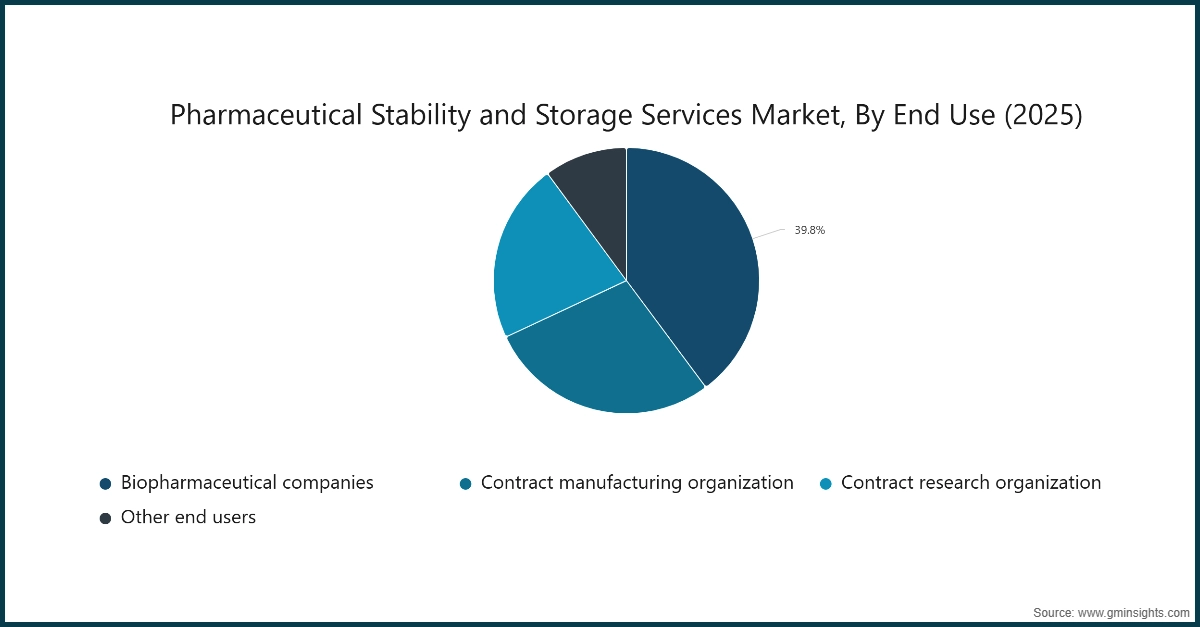
Based on end use, the pharmaceutical stability and storage services market is segmented into biopharmaceutical companies, contract manufacturing organization, contract research organization, and other end users. The biopharmaceutical companies segment held a dominating share of 39.8% in the market in 2025.
- Biopharmaceutical companies play an integral role in the development, production, and commercialization of a wide range of drugs. These companies invest heavily in ensuring regulatory compliance for stability testing and storage to maintain drug safety and efficacy throughout the product lifecycle.
- With the rapid growth of biologics, biosimilars, and innovative therapies, biopharmaceutical companies require advanced stability testing and precise storage solutions to meet stringent regulatory standards globally.
- Additionally, biopharmaceutical companies often manage extensive portfolios of small and large molecule drugs, which demand diverse stability studies and customized storage conditions.
- The continuous innovation in their drug pipelines and their direct engagement in clinical trials amplify the need for robust stability and storage services, reinforcing their dominant position in this market.
Looking for region specific data?
North America Pharmaceutical Stability and Storage Services Market
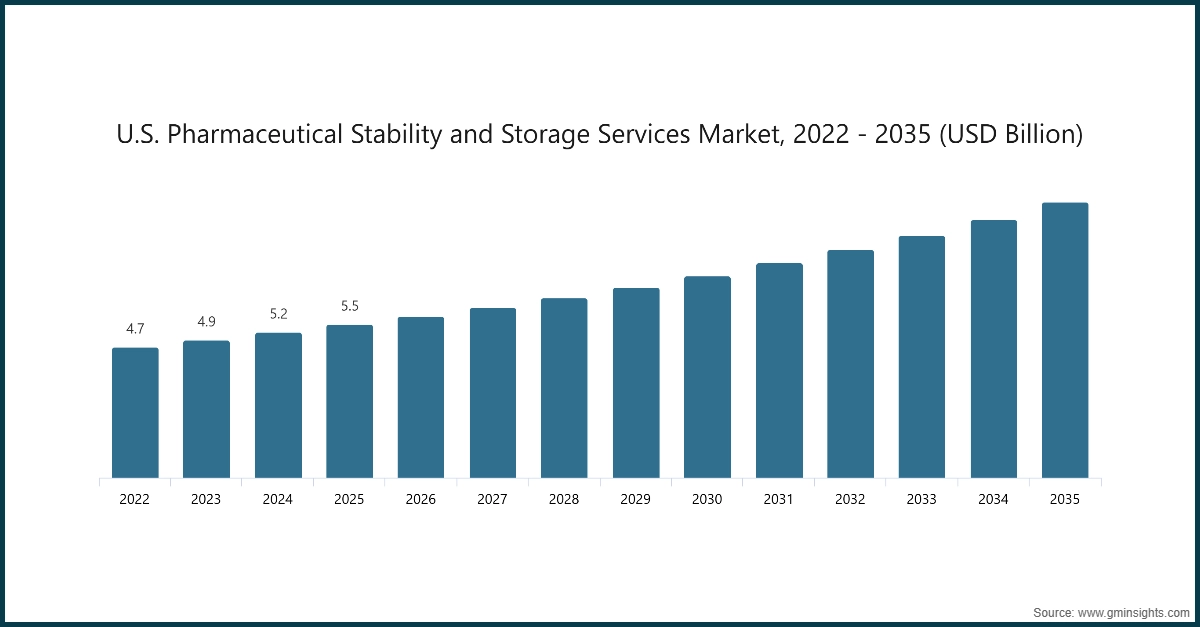
The North America pharmaceutical stability and storage services industry accounted for USD 6 billion in 2025 and is forecasted to reach USD 10.8 billion by 2035.
- North America accounted for the largest share of the market in 2025, driven by the region’s strong pharmaceutical manufacturing base, mature outsourcing ecosystem, and high reliance on validated stability testing and controlled-environment storage.
- The U.S. and Canada continue to dominate due to extensive drug development activity and stringent regulatory expectations requiring robust stability documentation.
- A well-established healthcare and pharmaceutical infrastructure, along with stringent FDA and Health Canada regulatory frameworks, reinforces North America’s leadership.
- These agencies impose rigorous stability testing and storage compliance requirements, prompting pharmaceutical and biotech companies to depend heavily on specialized service providers equipped with validated chambers, advanced monitoring systems, and high-quality environmental controls.
- With rising demand for outsourced stability testing, ongoing expansion of regional facilities, and continued pharmaceutical innovation, North America is expected to maintain its leading position throughout the forecast period.
The U.S. dominated the North America market with the largest revenue of USD 5.5 billion in 2025.
- In the U.S., the rapidly growing biologics demand is a significant driver of the pharmaceutical stability and storage services market. Biopharmaceutical companies require specialized storage solutions, such as refrigerated and frozen storage, to maintain the integrity of biologic products.
- With the U.S. being a leader in the production of biologics and biosimilars, the demand for stability services, including accelerated and photostability testing, is increasing.
- Another important factor driving market growth in the U.S. is the strict regulatory environment surrounding pharmaceutical manufacturing and storage. The U.S. Food and Drug Administration (FDA) mandates that pharmaceutical companies adhere to rigorous standards for stability testing, packaging, and storage to ensure the safety and efficacy of drugs.
- Compliance with the FDA’s Good Manufacturing Practices (GMP) guidelines require consistent stability and storage services to meet the necessary shelf-life requirements, contributing to the continued growth of the market in the country.
Europe Pharmaceutical Stability and Storage Services Market
Europe was valued at USD 4.3 billion in 2025 and is projected to witness steady growth over the forecast period.
- Europe holds a strong and influential position in the pharmaceutical stability and storage services industry, supported by its well‑established pharmaceutical manufacturing base, advanced regulatory framework, and robust research and development ecosystem. The region’s strict quality standards and regulatory oversight create sustained demand for validated stability testing environments and specialized storage facilities.
- Moreover, rising demand for outsourced stability storage services in Europe is driven by the growing burden of chronic diseases, expanding biologics and biosimilars pipelines, and increasingly complex drug formulations requiring controlled environmental conditions. As pharmaceutical innovation accelerates, the need for highly specialized stability infrastructure continues to expand.
- With strengthened emphasis on regulatory compliance, a mature pharmaceutical manufacturing landscape, and growing reliance on outsourced stability and storage services, Europe is expected to maintain a robust and expanding presence in the market throughout the forecast period.
The UK pharmaceutical stability and storage services market is expected to experience promising growth from 2026 to 2035.
- In the UK, the pharmaceutical sector’s focus on innovation and research has fueled the demand for stability and storage services. With a strong presence of pharmaceutical research organizations, particularly in Cambridge and London, stability testing is essential for the development of new drug formulations.
- The UK government’s ongoing investment in pharmaceutical R&D, including grants and funding for biopharmaceutical companies, supports the expansion of pharmaceutical stability services, which are critical for ensuring the quality and shelf life of novel drug products.
- Moreover, the UK’s post-Brexit regulatory changes have led to increased demand for stability and storage services as companies need to comply with both European Union and UK-specific regulations.
- Companies involved in drug manufacturing and distribution must ensure that their products meet the required stability standards to avoid market disruptions. The complexity of the UK’s dual-regulation system for the pharmaceutical industry is encouraging pharmaceutical companies to partner with specialized stability service providers, driving the growth of the market in the country.
Asia Pacific Pharmaceutical Stability and Storage Services Market
The Asia Pacific region is projected to exhibit lucrative growth of around 7.3% during the forecast period.
- Asia Pacific is emerging as one of the fastest-growing regions in the pharmaceutical stability and storage services industry, driven by the rapid expansion of the pharmaceutical and biopharmaceutical industries, increasing healthcare investment, and rising demand for reliable stability testing and controlled-environment storage.
- Countries such as China, India, Japan, South Korea, and Southeast Asian nations are contributing significantly to this momentum by strengthening drug development, manufacturing, and clinical research activities.
- A major catalyst for market growth in the region is the surge in large-molecule biologics, biosimilars, and temperature-sensitive therapeutics, which require stringent stability monitoring and specialized storage conditions. As pharmaceutical pipelines expand and regulatory expectations intensify, demand for sophisticated stability chambers, humidity-controlled rooms, and ultra-low-temperature storage is increasing across Asia Pacific.
- With accelerating pharmaceutical production, expanding biologics and biosimilars development, and growing demand for outsourced stability testing, Asia Pacific is expected to remain the fastest-growing regional market throughout the forecast period.
Japan pharmaceutical stability and storage services market is anticipated to witness lucrative growth between 2026 - 2035.
- In Japan, the increasing demand for advanced pharmaceuticals due to its aging population is significantly influencing the pharmaceutical stability and storage services industry.
- The country has one of the highest life expectancies in the world, which has led to a rise in chronic diseases and, consequently, an increase in the demand for pharmaceutical products.
- Additionally, Japan's rapid adoption of innovative drug delivery technologies, including novel biologics, gene therapies, and cell-based treatments, is contributing to the market growth.
- These products often require specialized storage conditions such as ultra-cold storage and cryogenic freezing to maintain their stability and effectiveness. As the demand for such advanced therapies increases, so does the need for pharmaceutical stability and storage services to meet stringent regulatory guidelines and maintain product quality during transportation and storage.
Latin America Pharmaceutical Stability and Storage Services Market
Brazil is experiencing significant growth in the pharmaceutical stability and storage services industry.
- Brazil is emerging as a key growth market within the global pharmaceutical stability and storage services landscape, underpinned by expanding drug development and manufacturing activity.
- Regulatory stringency is a structural accelerator of demand. ANVISA’s RDC 430/2020 sets good practices for distribution, storage, and transport of medicines, mandating temperature/humidity control, documentation, and risk‑based monitoring throughout the logistics chain, requirements that elevate the need for qualified stability chambers, continuous monitoring, and excursion management.
- The country’s demographic and healthcare investment patterns are supporting long‑term market expansion.
- Brazil is the largest pharmaceutical market in Latin America, and its universal healthcare system, widespread public health infrastructure, and increasing federal investments in science, technology, and biotechnology enhance the ecosystem for stability and storage services.
- With a large addressable base of pharmaceutical manufacturers, ongoing regulatory alignment with global standards, and the region’s continued expansion of outsourced stability testing, Brazil is expected to remain a strategically important market throughout the forecast period.
Middle East and Africa Pharmaceutical Stability and Storage Services Market
The pharmaceutical stability and storage services industry in Saudi Arabia is expected to experience lucrative growth from 2026 to 2035.
- In Saudi Arabia, the government’s Vision 2030 initiative, aimed at diversifying the economy, has spurred significant investments in the pharmaceutical and healthcare sectors.
- The government has sought to develop the pharmaceutical manufacturing industry, which has led to a need for stability and storage services to meet international standards.
- In addition to this, the extreme climate conditions in the country, with summer temperatures reaching as high as 40°C, have led to a need for temperature storage solutions.
- This has led to a surge in demand for refrigerated, frozen, and cryogenic storage solutions that meet SFDA standards, and this is expected to be driven by the growth of the pharmaceutical industry.
Pharmaceutical Stability and Storage Services Market Share
The pharmaceutical stability and storage services industry is highly competitive, driven by the growing need for GMP‑compliant stability studies, expansion of biologics pipelines, regulatory‑aligned storage solutions, and increasing outsourcing by pharmaceutical and biopharmaceutical companies. Leading service providers differentiate themselves through advanced stability testing technologies, global storage infrastructure, regulatory expertise, and the ability to support diverse molecule types across multiple ICH climatic zones.
Major global players such as Catalent, Charles River Laboratories, Eurofins Scientific, Intertek Group, and Almac Group consistently strengthen their market positions by investing in expanded storage capacity, technological innovation, and enhanced compliance frameworks. As innovation accelerates, particularly in biologics, biosimilars, cell and gene therapies, and precision‑medicine research, top providers are increasingly focused on ultra‑low‑temperature storage, real‑time monitoring technologies, validated stability chambers, and integrated analytical support. Their investments and expansion strategies position them as essential partners for biopharma companies seeking end‑to‑end outsourced solutions.
Pharmaceutical Stability and Storage Services Market Companies
Some of the eminent market participants operating in the pharmaceutical stability and storage services industry include:
- Alcami Corporation
- Almac Group
- Auriga Research
- Catalent
- Charles River Laboratories
- Element Materials Technology
- Eurofins Scientific
- Intertek Group
- Lucideon
- PD Partners
- Precision Stability Storage
- Q Laboratories
- Q1 Scientific
- Reading Scientific Services
- Roylance Stability Storage
- Eurofins Scientific
The company’s competitive strength comes from its ability to manage large volumes of stability samples across multiple climatic zones, its deep regulatory alignment, and its capacity to support global clinical development programs. Eurofins’ broad analytical capabilities allow it to seamlessly integrate stability programs with method validation, impurity profiling, and release testing.
Intertek’s advantage lies in its ability to deliver highly customized stability and storage solutions, leveraging advanced analytical technologies and robust quality systems. Its global reach enables pharmaceutical and biotech customers to conduct multi-site, multi-region stability studies with consistent data integrity and regulatory compliance.
Almac Group is a key provider of pharmaceutical stability and storage services, recognized for its specialized storage environments, comprehensive stability capabilities, and strong clinical supply chain integration. Industry assessments list Almac among the prominent companies shaping global capacity in stability and storage services.
Pharmaceutical Stability and Storage Services Market Report Attributes
| Key Takeaway | Details |
|---|---|
| Market Size & Growth | |
| Base Year | 2025 |
| Market Size in 2025 | USD 15.8 Billion |
| Market Size in 2026 | USD 16.8 Billion |
| Forecast Period 2026-2035 CAGR | 6.6% |
| Market Size in 2035 | USD 29.9 Billion |
| Key Market Trends | |
| Drivers | Impact |
| Growing focus on regulatory compliance | Increasing enforcement of GMP, GDP, and ICH stability guidelines is compelling pharmaceutical companies to outsource stability and storage functions to specialized service providers with validated systems, secure audit trails, and compliance-ready environments. |
| Increasing investments in drug development and research | The surge in the development of biologics, biosimilars, advanced therapies, and high-value specialty drugs is generating a higher volume of molecules requiring stability testing across various climatic zones. |
| Technological innovations enhancing pharmaceutical stability and storage | Advancements such as automated stability chambers, real-time condition monitoring, IoT-enabled temperature mapping, and AI-based predictive stability analytics improve reliability, reduce risks, and optimize resource efficiency |
| Expansion in global supply chains | Outsourced service providers enable manufacturers to efficiently meet region-specific regulatory requirements, maintain product integrity across long-distance transportation, and manage distributed clinical trial demands |
| Pitfalls & Challenges | Impact |
| High cost associated with specialized storage solutions | The need for temperature-controlled environments (e.g., 2–8°C, -20°C, -80°C), humidity-regulated chambers, backup power systems, and continuous monitoring significantly increase operational expenses for service providers. |
| Concerns related to transportation and logistics | Maintaining product stability during distribution requires robust cold-chain infrastructure, validated shipping systems, and strict monitoring of temperature excursions. |
| Opportunities: | Impact |
| Rising demand for end‑to‑end outsourced stability and storage ecosystems | Companies increasingly prefer integrated service providers offering protocol design, analytical testing, long-term storage, monitoring, and regulatory documentation, creating opportunities for full-suite stability service platforms. |
| Market Leaders (2025) | |
| Market Leader |
Market share of Catalent 15% |
| Top Players |
Top players collective market share 45% |
| Competitive Edge |
|
| Regional Insights | |
| Largest Market | North America |
| Fastest growing market | Asia Pacific |
| Emerging countries | India, China, Brazil, Mexico |
| Future outlook |
|
What are the growth opportunities in this market?
Pharmaceutical Stability and Storage Services Industry News:
- In July 2024, Catalent completed a USD 25 million expansion of its clinical supply facility in Schorndorf, Germany. This expansion added 32,000 square feet of new operational space, significantly increasing the site’s temperature-controlled storage capacity and introducing an advanced automatic bottle-filling line. This enhancement marks an important milestone for Catalent, strengthening its ability to support global clinical supply demands and reinforcing its position as a leading provider of pharmaceutical stability, storage, and clinical packaging services.
- In July 2024, Alcami Corporation introduced new conditions and expanded service capabilities at its state-of-the-art pharma storage facility in Garner, North Carolina. The 65,000-square-foot facility, strategically positioned near the Research Triangle Park (RTP) biotech and pharmaceutical hub, enhances Alcami’s ability to support growing client demand by offering expanded stability, cold-chain, and custom storage services. This development represents a significant milestone for the company.
The pharmaceutical stability and storage services market research report includes in-depth coverage of the industry with estimates and forecast in terms of revenue in USD Million from 2022 – 2035 for the following segments:
Market, By Service Type
- Stability
- Drug substance
- Stability indicating method validation
- Accelerated stability testing
- Photostability testing
- Other stability testing methods
- Storage
- Cold
- Frozen
- Refrigerated
- Controlled
- Cryogenic
- Non-cold
- Cold
Market, By Molecule Type
- Small molecule
- Commercial products
- Research products
- Large molecule
- Commercial products
- Research products
Market, By End Use
- Biopharmaceutical companies
- Contract manufacturing organization
- Contract research organization
- Other end users
The above information is provided for the following regions and countries:
- North America
- U.S.
- Canada
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Netherlands
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- Latin America
- Brazil
- Mexico
- Argentina
- Middle East and Africa
- Saudi Arabia
- South Africa
- UAE
Frequently Asked Question(FAQ) :
What was the market size of the pharmaceutical stability and storage services in 2025?
The market size was USD 15.8 billion in 2025, growing at a CAGR of 6.6% till 2035. The market is driven by increasing regulatory compliance, investments in drug R&D, and the expansion of global supply chains.
What is the projected value of the pharmaceutical stability and storage services market by 2035?
The market is poised to reach USD 29.9 billion by 2035, fueled by advancements in storage technologies and rising demand for stability testing services.
What is the expected size of the pharmaceutical stability and storage services industry in 2026?
The market size is expected to reach USD 16.8 billion in 2026.
What was the market share of the stability segment in 2025?
The stability segment dominated the market with a 60.8% share in 2025, led by the critical role of stability testing in ensuring drug safety and efficacy.
What was the market share of the small molecule segment in 2025?
The small molecule segment held a 54.7% market share in 2025, as small molecules are widely used in drug development and form the backbone of most oral medications.
What was the market share of biopharmaceutical companies in 2025?
Biopharmaceutical companies accounted for 39.8% of the market in 2025, reflecting their significant role in drug development, production, and regulatory compliance.
What is the growth outlook for the North America pharmaceutical stability and storage services sector?
The North America market, valued at USD 6 billion in 2025, is forecasted to reach USD 10.8 billion by 2035.
What are the upcoming trends in the pharmaceutical stability and storage services market?
Key trends include IoT-enabled real-time monitoring, advanced cryogenic storage technologies, automated robotic storage systems, and improved environmental monitoring to ensure compliance and reduce product degradation.
Who are the key players in the pharmaceutical stability and storage services industry?
Key players include Alcami Corporation, Almac Group, Auriga Research, Catalent, Charles River Laboratories, Element Materials Technology, Eurofins Scientific, Intertek Group, Lucideon, and PD Partners.
Pharmaceutical Stability and Storage Services Market Scope
Related Reports