Summary
Table of Content

Personalized Cancer Vaccine Market
Get a free sample of this report
Form submitted successfully!
Error submitting form. Please try again.
Thank you!
Your inquiry has been received. Our team will reach out to you with the required details via email. To ensure that you don't miss their response, kindly remember to check your spam folder as well!

Request Sectional Data
Thank you!
Your inquiry has been received. Our team will reach out to you with the required details via email. To ensure that you don't miss their response, kindly remember to check your spam folder as well!
Form submitted successfully!
Error submitting form. Please try again.
Personalized Cancer Vaccine Market Size
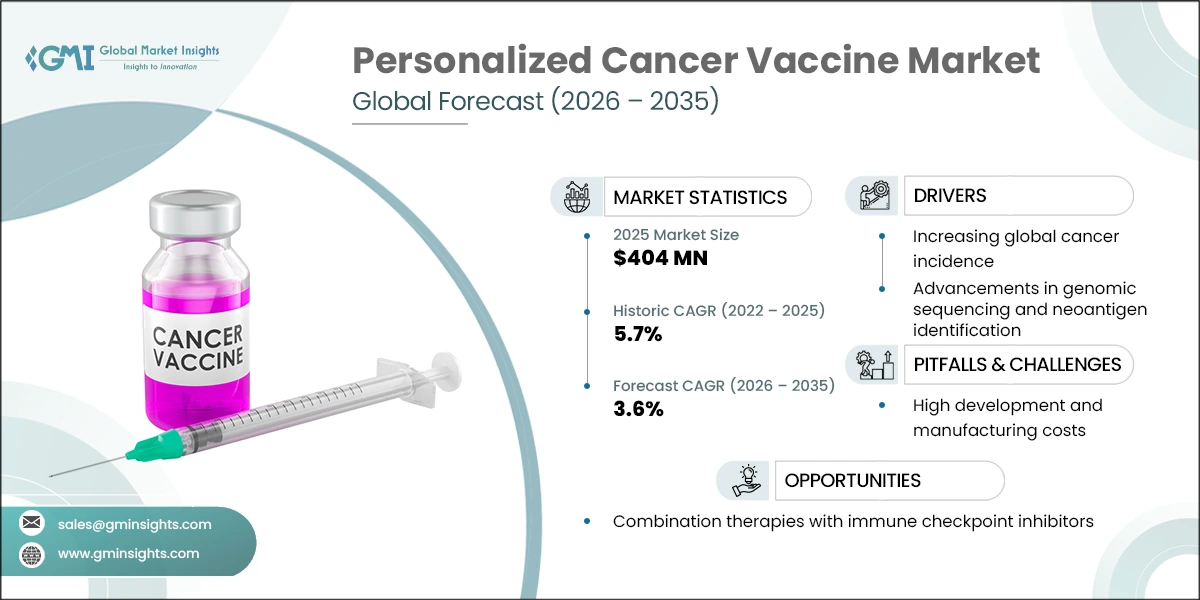
The global personalized cancer vaccine market was valued at USD 404 million in 2025, according to the latest report published by Global Market Insights Inc. The growth of the market can be attributed to the increasing cases of cancer and rising R&D in precision oncology research.
To get key market trends
According to the World Health Organization (WHO), cancer is a leading cause of death worldwide, accounting for nearly 10 million deaths in 2020, or nearly one in six deaths. In addition, the International Agency for Research on Cancer (IARC) estimates that the global number of cancer cases could reach over 28 million annually by 2040, reflecting a substantial rise in the disease burden and the need for advanced therapies such as personalized immunotherapies.
Personalized cancer vaccines are therapeutic vaccines designed to trigger an immune response against tumor-specific antigens or neoantigens that are unique to an individual patient’s cancer. These vaccines are developed using genomic sequencing of the patient’s tumor to identify mutations and generate customized antigens capable of activating T-cells that recognize and destroy cancer cells. The market currently includes only one approved personalized therapeutic cancer vaccine, sipuleucel-T (Provenge), which was approved by the U.S. FDA for the treatment of asymptomatic or minimally symptomatic metastatic castration-resistant prostate cancer (mCRPC).
Sipuleucel-T is an autologous cellular immunotherapy in which a patient’s immune cells are collected through leukapheresis, activated with a prostate cancer antigen (PAP-GM-CSF), and reinfused to stimulate a targeted immune response. Clinical trials supporting its approval demonstrated that the therapy improved median overall survival by approximately 4.1 months compared with placebo, with a 22% reduction in the risk of death. However, this product has faced several challenges, including high treatment costs, complex manufacturing processes, and modest clinical benefits, which limited widespread adoption.
Personalized Cancer Vaccine Market Report Attributes
| Key Takeaway | Details |
|---|---|
| Market Size & Growth | |
| Base Year | 2025 |
| Market Size in 2025 | USD 404 Million |
| Market Size in 2026 | USD 272.1 Million |
| Forecast Period 2026-2035 CAGR | 5.7% |
| Key Market Trends | |
| Drivers | Impact |
| Increasing global cancer incidence | The rising burden of cancer worldwide is a major driver for personalized cancer vaccines. According to the World Health Organization (WHO), over 35 million new cancer cases are predicted in 2050, a 77% increase from the estimated 20 million cases in 2022. This growing disease burden is increasing the demand for innovative therapies such as personalized vaccines that target tumor-specific antigens. |
| Advancements in genomic sequencing and neoantigen identification | Rapid improvements in next-generation sequencing (NGS), tumor profiling, and bioinformatics tools have enabled the identification of patient-specific tumor mutations and neoantigens. These technological advancements allow the development of highly individualized vaccines designed to stimulate precise immune responses against cancer cells. |
| Growing investment in cancer immunotherapy research | Governments, pharmaceutical companies, and research institutions are investing heavily in immuno-oncology. Increased funding for cancer research and the growing focus on precision medicine are accelerating the development of personalized vaccine platforms, including mRNA and peptide-based vaccines. |
| Expanding clinical pipeline for personalized cancer vaccines | Several biotechnology companies are actively developing personalized cancer vaccines across different technology platforms such as mRNA, peptide, DNA, and dendritic-cell vaccines. The growing number of clinical trials and late-stage pipeline candidates is expected to drive market growth over the coming years. |
| Pitfalls & Challenges | Impact |
| High development and manufacturing costs | Personalized cancer vaccines require patient-specific manufacturing processes. This complex process significantly increases development and production costs compared with conventional therapies. |
| Limited number of approved products | Currently, the personalized cancer vaccine market has very limited approved therapies, with sipuleucel-T (Provenge) being the only approved product. The limited commercialization of products restricts the market expansion. |
| Opportunities: | Impact |
| Combination therapies with immune checkpoint inhibitors | Combining personalized cancer vaccines with immune checkpoint inhibitors such as PD-1 or PD-L1 inhibitors has shown promising results in clinical trials. These combination approaches may improve treatment efficacy and expand the use of cancer vaccines. |
| Expansion into multiple cancer types | Current personalized cancer vaccine research is expanding beyond melanoma and prostate cancer into lung, pancreatic, colorectal, and breast cancers, significantly increasing the potential patient population. |
| Future outlook | The future growth potential of the personalized cancer vaccine market is highly promising., driven by rapid advances in precision medicine, mRNA technology, and tumor genomics.Increasing investment in cancer immunotherapy and the expanding pipeline of personalized vaccine candidates are expected to accelerate market growth over the next decade.Furthermore, as the number of clinical studies investigating personalized cancer vaccines continues to increase, the future growth potential of the personalized cancer vaccine market is highly promising. |
What are the growth opportunities in this market?
Personalized Cancer Vaccine Market Trends
- One of the most notable trends in the personalized cancer vaccine market is the rapid development of the mRNA-based vaccine platform. Several vaccine candidates based on the mRNA platform are in the clinical pipeline. for the treatment of melanoma, pancreatic cancer, and colorectal cancer.
- The development of mRNA vaccines has been a major breakthrough in the field of cancer therapy, with over 120 clinical trials conducted to date, demonstrating the potential of this technology in the treatment of various types of cancer, ranging from lung, breast, prostate, and melanoma cancers to other difficult-to-treat cancers like pancreatic and brain cancer.
- The pipeline for personalized cancer vaccines is rapidly expanding, particularly with the emergence of mRNA-based neoantigen vaccines. Several candidates are currently in late-stage clinical trials.
- One prominent example is mRNA-4157 (V940), developed by Moderna and Merck, which is a personalized mRNA vaccine designed to encode patient-specific neoantigens. In earlier clinical studies for melanoma, the vaccine in combination with KEYTRUDA (pembrolizumab) demonstrated a 44–49% reduction in the risk of recurrence or death compared with pembrolizumab alone, and the therapy has progressed to Phase III clinical trials.
- Another advanced candidate is Autogene cevumeran (BNT122), a personalized mRNA neoantigen vaccine being developed by BioNTech in collaboration with Genentech/Roche. It is designed to target specific tumor neoantigens and is currently being evaluated in Phase 2 trials for various solid tumors, including pancreatic, colorectal, and melanoma.
- These programs highlight the strong clinical interest in individualized immunotherapies and are expected to significantly expand the personalized cancer vaccine market once they are approved.
- Another key trend in the personalized cancer vaccine market is the growing funding and strategic investments aimed at accelerating research and clinical development. Pharmaceutical companies and governments are increasingly investing substantial capital to expand mRNA vaccine platforms, build manufacturing facilities, and support large-scale clinical trials.
- For instance, in May 2025, BioNTech announced its plans to invest up to USD 1.33 billion in the UK over the next ten years to broaden its research and development activities. Under the deal, BioNTech will enroll up to 10,000 patients in clinical trials by the end of 2030 for personalized cancer therapies and make investments to expand the company's footprint in the country.
- These growing investments are expected to accelerate technological innovation, expand clinical pipelines, and support the commercialization of next-generation personalized cancer vaccines in the coming years, thereby driving the market growth.
Personalized Cancer Vaccine Market Companies
Below is the list of prominent players in the personalized cancer vaccine market, including both approved/commercialized companies and emerging or pipeline-stage developers:
- Approved/commercialized players
- Dendreon Pharmaceuticals
- Emerging/ pipeline players
- Immatics
- Candel Therapeutics
- Evaxion Biotech
- Immunomic Therapeutics
- Imugene
- Elicio Therapeutics
- Infinitopes
- Takis Biotech
- VacV Biotherapeutics
- BioNTech
- Moderna
- OSE Immunotherapeutics
- ImmunityBio
- Agenus
Dendreon Pharmaceuticals is a key player in the personalized cancer vaccine market owing to its product sipuleucel-T (Provenge), the first therapeutic cancer vaccine approved by the U.S. Food and Drug Administration (FDA). Provenge, approved in 2010, is an autologous active cellular immunotherapy and has shown evidence of efficacy in reducing the risk of death among men with metastatic castration-resistant prostate cancer.
Phase III trials (IMPACT) showed that Provenge reduces the risk of death by 22%, increasing median overall survival by 4.1 months compared to placebo, establishing it as the first personalized immunotherapy to show a survival benefit in advanced prostate cancer. Since its FDA-approval in 2010, nearly 40,000 men have been prescribed Provenge.
Moderna is a prominent player in the personalized cancer vaccine market, leveraging its messenger RNA (mRNA) technology platform to develop individualized immunotherapies targeting tumor-specific mutations. The company is actively advancing mRNA-4157 (also known as V940), a personalized neoantigen mRNA cancer vaccine designed to encode multiple patient-specific tumor antigens that are identified through genomic sequencing. This vaccine is developed in collaboration with Merck & Co., Inc. and is being evaluated in combination with pembrolizumab for several cancers, including melanoma and non-small cell lung cancer. Clinical trial results have demonstrated promising efficacy, with studies showing that the combination therapy has reduced the risk of recurrence or death in high-risk melanoma patients by approximately 49% compared with pembrolizumab alone.
Personalized Cancer Vaccine Industry News
- In January 2026, Moderna, Inc. and Merck & Co., Inc. reported long-term data indicating that their personalized cancer vaccine mRNA-4157 (V940) combined with pembrolizumab reduced the risk of recurrence or death in melanoma patients by approximately 49%. Positive long-term efficacy results enhance the companies’ credibility in immuno-oncology and support the potential commercialization of personalized cancer vaccines.
- In November 2024, Transgene S.A. and NEC Corporation presented clinical data demonstrating that their personalized neoantigen cancer vaccine TG4050 generated strong immune responses in patients with head and neck cancer. This advancement helps strengthen the companies’ clinical development pipeline and highlights the potential of their AI-driven neoantigen identification platform.
- In October 2024, Moderna, Inc. and Merck & Co., Inc. announced the initiation of INTerpath-009, a pivotal Phase 3 randomized clinical trial evaluating V940 (mRNA-4157) in combination with KEYTRUDA (pembrolizumab) for patients with resectable non-small cell lung cancer and melanoma. This development strengthens the companies’ oncology pipelines and positions them as leading players in the emerging personalized cancer vaccine market.
- In May 2024, BioNTech SE announced positive Phase 2 clinical data for its mRNA cancer vaccine candidate BNT111, developed for advanced melanoma, in combination with cemiplimab. The study demonstrated promising anti-tumor activity and durable immune responses in patients with anti-PD-1-refractory melanoma. These findings support the continued advancement of BioNTech’s FixVac vaccine platform and strengthen the company’s oncology vaccine pipeline.
- In April 2024, Gritstone bio, Inc. announced positive preliminary data from the ongoing, signal-seeking Phase 2 portion of the Phase 2/3 study evaluating GRANITE, its personalized neoantigen cancer vaccine, in front-line metastatic microsatellite-stable colorectal cancer. This advancement supports the company’s strategy to develop next-generation individualized cancer vaccines and expand its presence in precision immunotherapy.
The personalized cancer vaccine market research report includes in-depth coverage of the industry with estimates and forecast in terms of revenue in USD Million from 2022 - 2035 for the following segments:
Market sizing analysis
- Approved product market assessment – U.S.
- Future market potential analysis (Pipeline-driven projection) - Global
- Total addressable market (TAM) - Global
Frequently Asked Question(FAQ) :
What is the market size of the personalized cancer vaccine market in 2025?
The global personalized cancer vaccine market was valued at USD 404 million in 2025.
What is the projected growth rate of the industry from 2026 to 2035?
The market is expected to grow at a forecast CAGR of 3.6% during the period of 2026 – 2035.
How many personalized cancer vaccines are currently approved?
Currently, the market has only one approved product: sipuleucel-T (Provenge), used for treating metastatic castration-resistant prostate cancer.
What are the primary drivers for this market?
Key drivers include increasing global cancer incidence, advancements in genomic sequencing, and growing investment in immuno-oncology research.
What are the significant challenges facing the market?
The industry faces high development and manufacturing costs due to patient-specific processes and a limited number of approved products.
Who are the prominent players in the personalized cancer vaccine industry?
Leading companies include Dendreon Pharmaceuticals (approved), as well as pipeline developers like Moderna, BioNTech, Immatics, and Merck & Co., Inc.
Personalized Cancer Vaccine Market Scope
Related Reports