Summary
Table of Content

Atrial Fibrillation Devices Market
Get a free sample of this report
Form submitted successfully!
Error submitting form. Please try again.
Thank you!
Your inquiry has been received. Our team will reach out to you with the required details via email. To ensure that you don't miss their response, kindly remember to check your spam folder as well!

Request Sectional Data
Thank you!
Your inquiry has been received. Our team will reach out to you with the required details via email. To ensure that you don't miss their response, kindly remember to check your spam folder as well!
Form submitted successfully!
Error submitting form. Please try again.
Atrial Fibrillation Devices Market Size
The market growth is attributed to the ongoing advancements in atrial fibrillation (AF) devices, rising prevalence of atrial fibrillation, increasing adoption of cardiac ablation procedures, and growing awareness and early diagnosis, among other contributing factors. Atrial fibrillation devices are medical tools designed to diagnose, monitor, and treat atrial fibrillation (AFib), a common heart rhythm disorder. These devices include catheters for ablation procedures, implantable cardiac monitors, pacemakers, and defibrillators that regulate heart rhythm. They aim to restore normal heart function, prevent stroke, and reduce AFib-related symptoms. These devices play a crucial role in both acute and long-term management of AFib patients.
Atrial Fibrillation Devices Market Key Takeaways
Market Size & Growth
- 2025 Market Size: USD 12.9 Billion
- 2026 Market Size: USD 14.3 Billion
- 2035 Forecast Market Size: USD 40.2 Billion
- CAGR (2026–2035): 12.2%
Regional Dominance
- Largest Market: North America
- Fastest Growing Region: Asia Pacific
Key Market Drivers
- High adoption rate of advanced technologies and presence of sophisticated healthcare infrastructure.
- Increase in prevalence of cardiovascular diseases.
- Favorable reimbursement scenario.
- Rise in prevalence of rheumatic valvular heart diseases.
Challenges
- High cost associated with atrial fibrillation devices.
- Lack of awareness regarding enhanced medical technologies in developing countries.
Opportunity
- Expansion of minimally invasive and catheter‑based therapies.
Key Players
- Market Leader: Abbott led with over 15% market share in 2025.
- Leading Players: Top 5 players in this market include Abbott, Boston Scientific Corporation, Johnson and Johnson, Medtronic, Stryker Corporation, which collectively held a market share of 35% in 2025.
Get Market Insights & Growth Opportunities
The key players in the atrial fibrillation devices market are Abbott, Boston Scientific Corporation, Johnson and Johnson, Medtronic, and Stryker Corporation. These players compete in the market through continuous product innovation, strategic mergers and acquisitions, geographic expansion, and investments in advanced electrophysiology and catheter ablation technologies, while also focusing on clinical collaborations and regulatory approvals to strengthen their global market presence.
The market increased from USD 9.6 billion in 2022 to USD 11.7 billion in 2024, with a historic growth rate of 10.6%. The market has shown steady expansion in recent years, supported by the rising global prevalence of atrial fibrillation, growing geriatric population, and increasing adoption of minimally invasive catheter ablation procedures.
The increasing incidence of atrial fibrillation worldwide is driving the market growth positively. AF is the most common type of cardiac arrhythmia, and its prevalence is rising due to factors like aging populations, increasing rates of obesity, hypertension, and other cardiovascular conditions. For instance, according to the Centers for Disease Control and Prevention (CDC), in 2023, AFib affects about 40 million people globally, including 6 million in the U.S. alone. Additionally, it is estimated that around 12.1 million people in the U.S. would have AFib by 2030. Hence, the growing number of AF cases leads to higher demand for diagnostic and treatment devices.
Furthermore, increased awareness of atrial fibrillation among both patients and healthcare providers has led to earlier diagnosis and treatment. Public health initiatives and educational campaigns emphasize the importance of monitoring and managing AF, which, in turn, boosts the demand for AF devices. Early detection and intervention are crucial in preventing complications like stroke, leading to greater use of diagnostic and monitoring devices.
The global atrial fibrillation devices market was valued at USD 12.9 billion in 2025. The market is expected to grow from USD 14.3 billion in 2026 to USD 40.2 billion in 2035, growing at a CAGR of 12.2% during the forecast period, according to the latest report published by Global Market Insights Inc.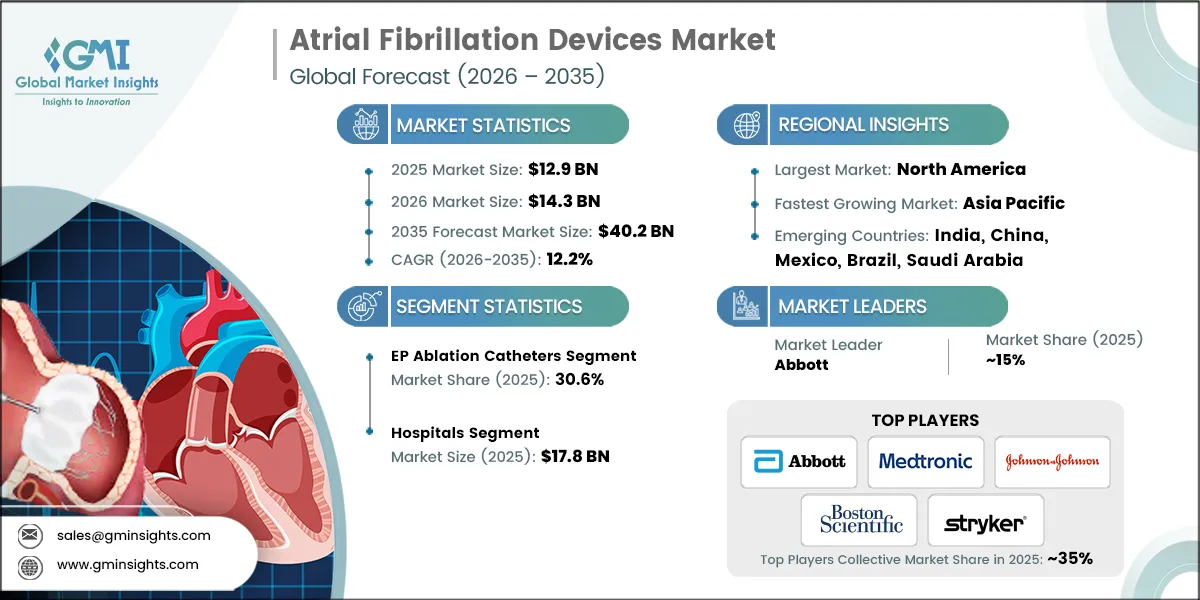
To get key market trends
Atrial Fibrillation Devices Market Trends
The market is experiencing several notable trends that are shaping its growth and development. Factors such as continuous innovations in technology, growing demand for minimally invasive procedures, growing adoption of wearable and remote monitoring devices, and increasing use of hybrid procedures, among other factors, are propelling the industry growth.
- One of the most significant trends in the atrial fibrillation (AF) devices market is the growing preference for minimally invasive procedures. Catheter ablation, which involves using heat or cold to destroy the tissue causing irregular heartbeats, is becoming increasingly popular due to its effectiveness and quicker recovery times. This trend is driving the development and adoption of advanced ablation catheters and other minimally invasive devices, making AF treatments less invasive and more patient-friendly.
- Moreover, with the rise of digital health, there is an increasing trend towards the use of wearable devices and remote monitoring systems for managing atrial fibrillation. These devices allow continuous monitoring of heart rhythm, enabling early detection of AF episodes and timely intervention. The integration of artificial intelligence (AI) and machine learning algorithms into these devices is further enhancing their diagnostic capabilities, leading to better patient outcomes and more proactive management of AF. These factors are expected to drive the market.
Atrial Fibrillation Devices Market Analysis
Learn more about the key segments shaping this market
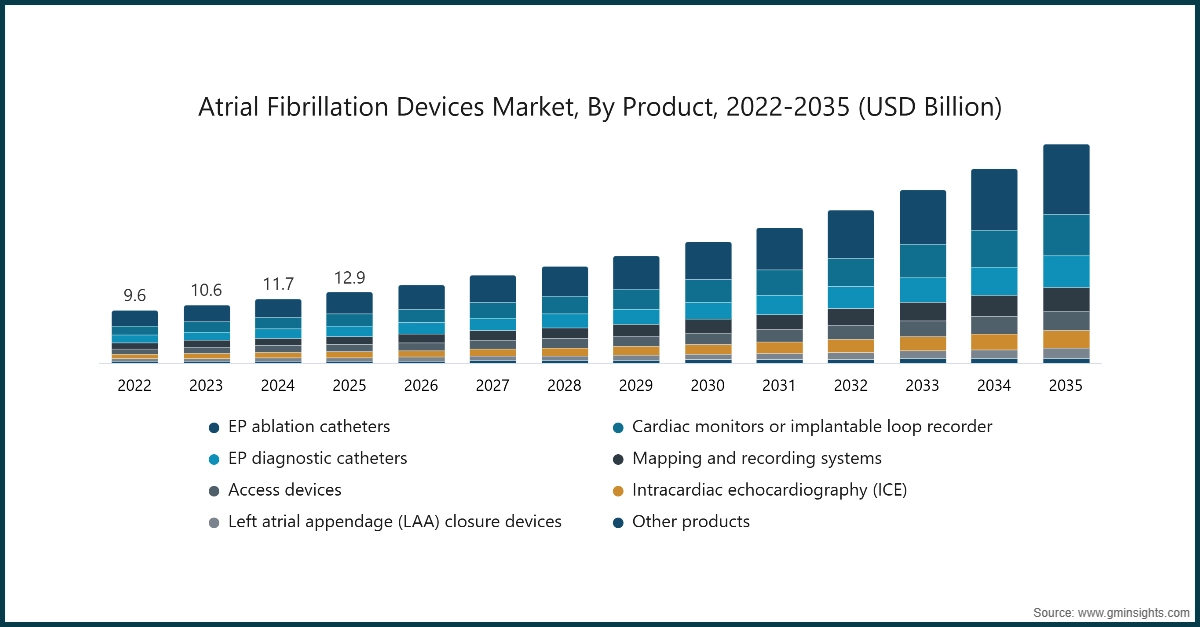
Based on product, the market is segmented into EP ablation catheters, cardiac monitors or implantable loop recorder, EP diagnostic catheters, mapping and recording systems, access devices, intracardiac echocardiography (ICE), left atrial appendage (LAA) closure devices, and other products. The EP ablation catheters segment has asserted its dominance in the market by securing a significant market share of 30.6% in 2025, owing to their critical role in catheter-based ablation procedures for the treatment of atrial fibrillation and other cardiac arrhythmias. These catheters enable precise tissue targeting, improved lesion formation, and enhanced procedural success rates, making them a preferred choice among electrophysiologists. The segment is expected to exceed USD 12.9 billion by 2035, growing at a CAGR of 12.7% during the forecast period.
- Technological advancements in EP ablation catheters, such as contact force-sensing technology, irrigated tip catheters, and improved energy delivery systems, are enhancing the safety and efficacy of ablation procedures. These innovations are making ablation a more viable option for a wider range of patients, thereby driving market growth. Improved catheter designs are also reducing procedure times and increasing the success rates of AF treatments.
- Further, there is a growing demand for minimally invasive procedures in healthcare, and ablation catheters align with this trend. Ablation procedures are less invasive compared to traditional surgical approaches and offer quicker recovery times, lower risks of complications, and shorter hospital stays. This preference for minimally invasive options is boosting the demand for EP ablation catheters in the AF devices market.
- The cardiac monitors or implantable loop recorder segment was valued at USD 2.3 billion in 2025 and is expected to reach USD 7.7 billion by 2035. This growth is primarily driven by the increasing prevalence of intermittent and asymptomatic atrial fibrillation cases that require long-term rhythm monitoring for accurate diagnosis and management. Implantable loop recorders offer continuous cardiac monitoring over extended periods, improving arrhythmia detection rates and enabling timely clinical intervention.
- The EP diagnostic catheters segment was valued at USD 1.9 billion in 2025 and is expected to reach USD 5.7 billion by 2035, driven by the growing number of electrophysiology procedures for precise arrhythmia mapping and diagnosis. Increasing adoption of minimally invasive cardiac interventions and technological advancements in high-resolution mapping systems are further supporting segment growth.
- The mapping and recording systems segment was valued at USD 1.5 billion in 2025 and is expected to reach USD 4.3 billion by 2035, driven by increasing demand for advanced 3D cardiac mapping technologies that enhance procedural accuracy and clinical outcomes in atrial fibrillation treatment. Continuous innovations in real-time imaging, integration with ablation systems, and rising electrophysiology lab installations are further accelerating segment growth.
- The access devices segment was valued at USD 1.3 billion in 2025 and is expected to reach USD 3.6 billion by 2035, supported by the rising volume of electrophysiology procedures and the need for reliable vascular access solutions during catheter-based interventions. Increasing adoption of minimally invasive cardiac treatments, along with advancements in steerable sheaths and introducer technologies, is further contributing to segment expansion.
- The intracardiac echocardiography (ICE) segment was valued at USD 1.1 billion in 2025 and is expected to reach USD 3.3 billion by 2035, driven by its growing use in real-time visualization during complex electrophysiology and ablation procedures. The technology enhances procedural precision, reduces fluoroscopy exposure, and improves clinical outcomes, which is accelerating adoption across advanced cardiac care centers.
- The left atrial appendage (LAA) closure devices segment was valued at USD 624.2 million in 2025 and is expected to reach USD 1.8 billion by 2035, supported by the rising preference for minimally invasive stroke prevention therapies in atrial fibrillation patients who are unsuitable for long-term anticoagulation. Increasing clinical evidence, expanding physician awareness, and ongoing product innovations are further contributing to the segment’s steady growth trajectory.
- The other product segment was valued at USD 308 million in 2025 and is expected to reach USD 820.1 million by 2035, driven by the growing adoption of adjunct electrophysiology tools and supportive technologies that enhance procedural efficiency and patient outcomes. Continuous technological refinements, along with increasing procedural volumes and expanding electrophysiology lab capabilities, are expected to sustain segment growth over the forecast period.
Learn more about the key segments shaping this market
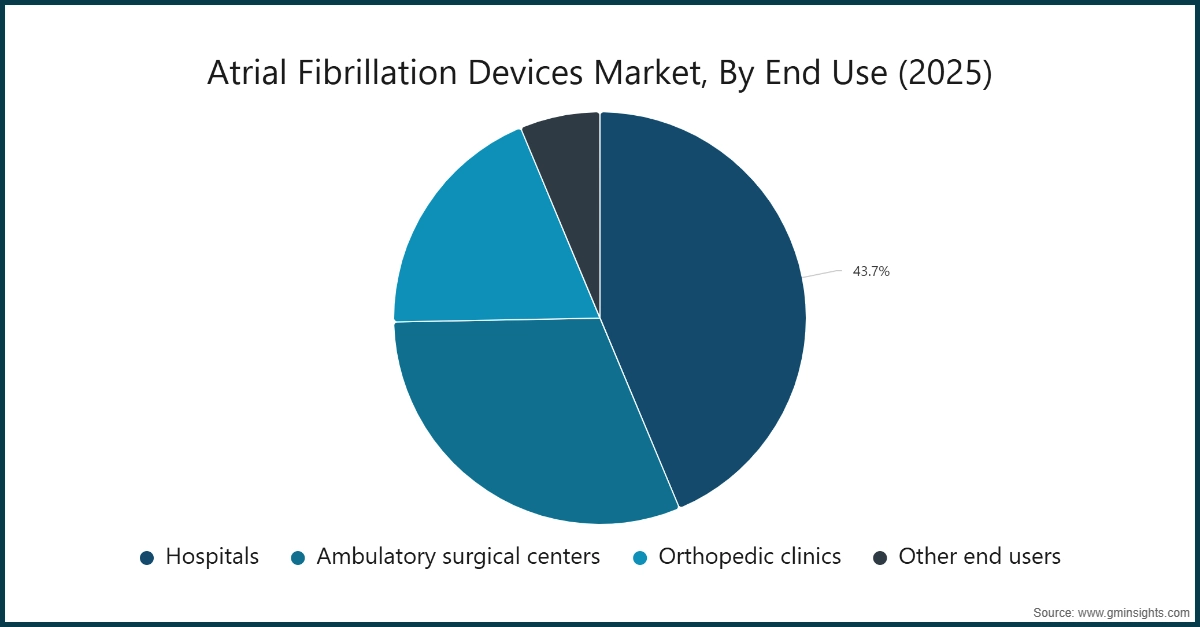
Based on end use, the atrial fibrillation devices market is segmented into hospitals, ambulatory surgical centers, cardiac centers, and other end users. The hospitals segment dominated the market with a revenue share of 43.7% in 2025 and is expected to reach USD 17.8 billion within the forecast period.
- The growing demand for specialized treatment centers dedicated to managing atrial fibrillation within hospitals is driving investment in AF devices. These centers require a wide range of devices for diagnosis, treatment, and patient monitoring, leading to an increase in device procurement by hospitals.
- Furthermore, many hospitals are seeking accreditation for excellence in cardiac care, which often requires the adoption of the latest AF treatment technologies. Accreditation and quality improvement initiatives drive hospitals to invest in advanced atrial fibrillation devices, contributing to market growth.
- The cardiac centers segment was valued at USD 4 billion in 2025 and is expected to reach USD 12 billion by 2035, growing at a CAGR of 11.8% during the forecast period. This growth is primarily attributed to the increasing number of specialized electrophysiology procedures performed in dedicated cardiac centers, supported by advanced infrastructure, skilled cardiologists, and improved patient referral networks. Additionally, rising awareness of minimally invasive atrial fibrillation treatments and expanding investments in cardiac care facilities are expected to further accelerate segment expansion over the forecast period.
- The ambulatory surgical centers segment was valued at USD 2.5 billion in 2025 and is expected to reach USD 8.1 billion by 2035. The segment’s growth is driven by the increasing shift toward minimally invasive electrophysiology and ablation procedures performed in outpatient settings, offering reduced hospital stays and lower procedural costs.
- The other end users segment is expected to witness steady growth over the forecast period. This segment, which includes specialty clinics and research institutes, is expanding due to the rising adoption of advanced diagnostic and monitoring devices for early atrial fibrillation detection and follow-up care.
Looking for region specific data?
North America Atrial Fibrillation Devices Market
North America dominated the global market, accounting for a market share of 40.6% in 2025.
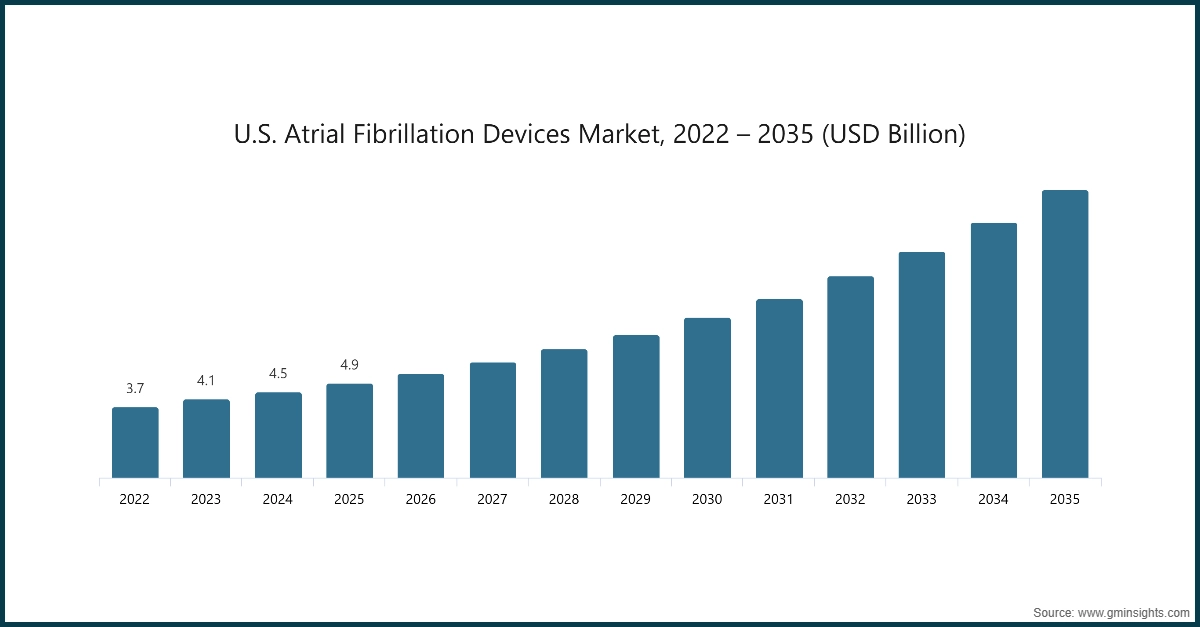
- The U.S. market was valued at USD 3.7 billion and USD 4.1 billion in 2022 and 2023, respectively. The market size reached USD 4.9 billion in 2025, growing from USD 4.5 billion in 2024, and is anticipated to grow at a CAGR of 12% from 2026 to 2035.
- North America, particularly the U.S., has a high prevalence of atrial fibrillation, driven by an aging population, increasing incidence of lifestyle-related conditions such as obesity, hypertension, and diabetes, and a rise in overall cardiovascular disease. This growing AF patient pool drives the demand for devices that can effectively diagnose and treat the condition, including ablation catheters, pacemakers, and diagnostic tools.
- Furthermore, the region’s high healthcare expenditure enables significant investments in advanced medical technologies, including atrial fibrillation devices. The willingness of healthcare providers to invest in cutting-edge treatments and the high level of healthcare spending per capita further stimulate the market, leading to regional growth.
Europe Atrial Fibrillation Devices Market
The Europe market accounted for USD 3.6 billion in 2025 and is anticipated to show lucrative growth over the forecast period.
- Europe represents a significant share of the market, supported by a high prevalence of cardiovascular diseases and a rapidly aging population. Countries such as Germany, France, the UK, and Italy are witnessing increased adoption of catheter ablation procedures and advanced electrophysiology mapping systems.
- Favorable reimbursement policies, well-established cardiac care infrastructure, and strong presence of leading medical device manufacturers are further strengthening regional growth. Additionally, increasing awareness of minimally invasive stroke prevention solutions, including LAA closure devices, is contributing to steady market expansion across the region.
Asia Pacific Atrial Fibrillation Devices Market
The Asia Pacific market is anticipated to grow at the highest CAGR of 13.2% during the forecast period.
- The Asia Pacific market is expected to register the fastest growth during the forecast period, driven by the rising burden of atrial fibrillation, improving healthcare infrastructure, and expanding access to advanced cardiac treatments.
- Rapid urbanization, increasing healthcare expenditure, and growing medical tourism in countries such as China, India, and Japan are accelerating demand for electrophysiology procedures.
- Furthermore, government initiatives aimed at enhancing cardiac care facilities and the increasing penetration of global device manufacturers are supporting robust regional growth.
Latin America Atrial Fibrillation Devices Market
The Latin America market accounted for USD 725.9 million in 2025 and is anticipated to show lucrative growth over the forecast period.
- The Latin America market is experiencing gradual growth, primarily driven by improving access to specialized cardiac care and rising awareness of arrhythmia management. Brazil and Mexico are leading contributors due to expanding healthcare investments and increasing adoption of minimally invasive cardiac interventions.
- Although reimbursement challenges and cost constraints remain, growing private healthcare participation and the establishment of advanced cardiac centers are expected to positively influence market development over the forecast period.
Middle East and Africa Atrial Fibrillation Devices Market
The Middle East and Africa market accounted for USD 357.9 million in 2025 and is anticipated to show lucrative growth over the forecast period.
- The MEA region is anticipated to witness steady growth, supported by rising cardiovascular disease prevalence and increasing investments in healthcare infrastructure.
- Nations such as Saudi Arabia and the UAE are expanding advanced cardiac treatment capabilities and adopting innovative electrophysiology technologies.
- While parts of Africa face limitations related to affordability and access to specialized care, improving healthcare reforms and growing public-private partnerships are expected to gradually enhance market penetration across the region.
Atrial Fibrillation Devices Market Share
The market is highly dynamic, with key players like Abbott Laboratories, Boston Scientific Corporation, and Medtronic, which collectively hold approximately 35% of the total market share. These players lead the market through innovative technologies such as ablation catheters, implantable devices, and advanced monitoring tools. Intense competition drives continuous research and development, resulting in more effective and minimally invasive treatment options. Emerging companies and startups are also gaining traction by introducing novel solutions. Strategic partnerships, acquisitions, and product launches are common strategies used to maintain market leadership. Additionally, regulatory approvals and reimbursement policies play a crucial role in shaping market competition and growth
Atrial Fibrillation Devices Market Companies
A few of the prominent players operating in the atrial fibrillation devices industry include:
- Abbott
- Acutus Medical
- AtriCure
- Biotronik
- Boston Scientific Corporation
- CardioFocus
- CathRx
- Hansen Medical
- Imricor
- Johnson and Johnson
- Medtronic
- MicroPort Scientific Corporation
- OSYPKA MEDICAL
- Stryker Corporation
- Synaptic Medical
- Abbott
Abbott stands out with its Volt pulsed‑field ablation (PFA) system, which combines mapping, pacing, and ablation in one device, offering doctors a simpler and faster AF treatment option backed by strong safety and effectiveness results across both paroxysmal and persistent AF.
Boston Scientific holds a strong first‑mover advantage in pulsed‑field ablation with its FARAPULSE system, one of the most widely used PFA platforms globally, supported by extensive real‑world data showing high procedural safety, rapid treatments, and strong long‑term effectiveness.
Johnson & Johnson leverages decades of leadership in electrophysiology and its fully integrated CARTO 3 mapping ecosystem to deliver AF solutions that work together seamlessly, improving precision, navigation, and overall treatment consistency for clinicians.
Atrial Fibrillation Devices Market Report Attributes
| Key Takeaway | Details |
|---|---|
| Market Size & Growth | |
| Base Year | 2025 |
| Market Size in 2025 | USD 12.9 Billion |
| Market Size in 2026 | USD 14.3 Billion |
| Forecast Period 2026-2035 CAGR | 12.2% |
| Market Size in 2035 | USD 40.2 Billion |
| Key Market Trends | |
| Drivers | Impact |
| High adoption rate of advanced technologies and presence of sophisticated healthcare infrastructure | Accelerates the uptake of newer AF devices, improving treatment efficiency and boosting overall market growth. |
| Increase in prevalence of cardiovascular diseases | Expands the patient pool requiring AF diagnosis and intervention, driving sustained demand for AF devices. |
| Favorable reimbursement scenario | Enhances patient access to AF therapies, increasing procedure volumes and device utilization. |
| Rise in prevalence of rheumatic valvular heart diseases | Leads to a higher incidence of AF complications, creating a greater need for monitoring and surgical device solutions. |
| Pitfalls & Challenges | Impact |
| High cost associated with atrial fibrillation devices | Limits adoption among cost‑sensitive healthcare facilities and patients, especially in low‑income segments. |
| Lack of awareness regarding enhanced medical technologies in developing countries | Slows market penetration and reduces uptake of advanced AF treatment options in emerging regions. |
| Opportunities: | Impact |
| Expansion of minimally invasive and catheter‑based therapies | Creates strong growth potential as patients and clinicians increasingly prefer safer, faster, and more effective AF treatment methods. |
| Market Leaders (2025) | |
| Market Leader |
Market share ~15% |
| Top Players |
Collective market share ~35% |
| Competitive Edge |
|
| Regional Insights | |
| Largest Market | North America |
| Fastest growing market | Asia Pacific |
| Emerging countries | India, China, Mexico, Brazil, Saudi Arabia |
| Future outlook |
|
What are the growth opportunities in this market?
Atrial Fibrillation Devices Industry News:
- In October 2024, Boston Scientific announced that it had received FDA clearance for its ablation catheter, which is equipped with navigation capabilities to be used for the treatment of paroxysmal AFib, along with 510(k) clearance for fresh imaging software for its pulsed field ablation system. The clearance would serve to strengthen the company's product line and thus enable it to provide more accurate and effective treatment solutions, thus expanding its position.
- In October 2024, Medtronic plc announced FDA approval for the Affera Mapping and Ablation System with Sphere-9 Catheter, a blended high-density mapping and pulsed field and radiofrequency ablation catheter for the treatment of persistent AFib and cavotricuspid isthmus-dependent atrial flutter. The approval was anticipated to strengthen Medtronic's portfolio, providing sophisticated solutions for arrhythmia therapy and solidifying its position in the market.
The atrial fibrillation devices market research report includes an in-depth coverage of the industry with estimates and forecasts in terms of revenue in USD Million from 2022 – 2035 for the following segments:
Market, By Product
- EP ablation catheters
- Radiofrequency (RF)
- Laser
- Cryoablation
- Ultrasound
- Microwave
- Other EP ablation catheters
- Cardiac monitors or implantable loop recorder
- EP diagnostic catheters
- Advanced mapping catheters
- Steerable catheters
- Fixed curve catheters
- Mapping and recording systems
- Access devices
- Intracardiac echocardiography (ICE)
- Left atrial appendage (LAA) closure devices
- Other products
Market, By End Use
- Hospitals
- Cardiac centers
- Ambulatory surgical centers
- Other end-users
The above information is provided for the following regions and countries:
- North America
- U.S.
- Canada
- Europe
- Germany
- UK
- France
- Spain
- Italy
- Netherlands
- Asia Pacific
- China
- Japan
- India
- Australia
- South Korea
- Latin America
- Brazil
- Mexico
- Argentina
- Middle East and Africa
- South Africa
- Saudi Arabia
- UAE
Frequently Asked Question(FAQ) :
What is the market size of the atrial fibrillation devices market in 2025?
The global market size for atrial fibrillation devices was valued at USD 12.9 billion in 2025 and is projected to grow significantly due to the rising prevalence of atrial fibrillation.
What is the market size of the atrial fibrillation devices market in 2026?
The market is expected to reach USD 14.3 billion in 2026, driven by growing awareness of early atrial fibrillation diagnosis.
What is the projected value of the atrial fibrillation devices market by 2035?
The market is projected to reach USD 40.2 billion by 2035, growing at a CAGR of 12.2% during the forecast period due to technological innovations.
Which product segment dominated the market in 2025?
The EP ablation catheters segment dominated the market with a 30.6% share in 2025, driven by the growing adoption of catheter-based ablation procedures for effective treatment of atrial fibrillation.
What was the valuation of the cardiac monitors or implantable loop recorder segment in 2025?
The cardiac monitors or implantable loop recorder segment was valued at USD 2.3 billion in 2025 and is expected to reach USD 7.7 billion by 2035, supported by the need for long-term heart rhythm monitoring.
Which region leads the atrial fibrillation devices industry?
North America led the market with a 40.6% share in 2025, supported by advanced healthcare infrastructure, high prevalence of atrial fibrillation, and strong presence of major medical device manufacturers.
How large was the Europe atrial fibrillation devices market in 2025?
The Europe atrial fibrillation devices market was valued at USD 3.6 billion in 2025, driven by increasing adoption of catheter ablation procedures and strong cardiac care infrastructure across countries such as Germany, France, and the UK.
Who are the key players in the atrial fibrillation devices market?
Key players include Abbott, Boston Scientific, Johnson & Johnson, Medtronic, and Stryker Corporation, which compete through product innovation, strategic partnerships, and investments in advanced electrophysiology technologies.
Atrial Fibrillation Devices Market Scope
Related Reports