Summary
Table of Content

Lyophilized Injectable Drugs Market
Get a free sample of this report
Form submitted successfully!
Error submitting form. Please try again.
Thank you!
Your inquiry has been received. Our team will reach out to you with the required details via email. To ensure that you don't miss their response, kindly remember to check your spam folder as well!

Request Sectional Data
Thank you!
Your inquiry has been received. Our team will reach out to you with the required details via email. To ensure that you don't miss their response, kindly remember to check your spam folder as well!
Form submitted successfully!
Error submitting form. Please try again.
Lyophilized Injectable Drugs Market Size
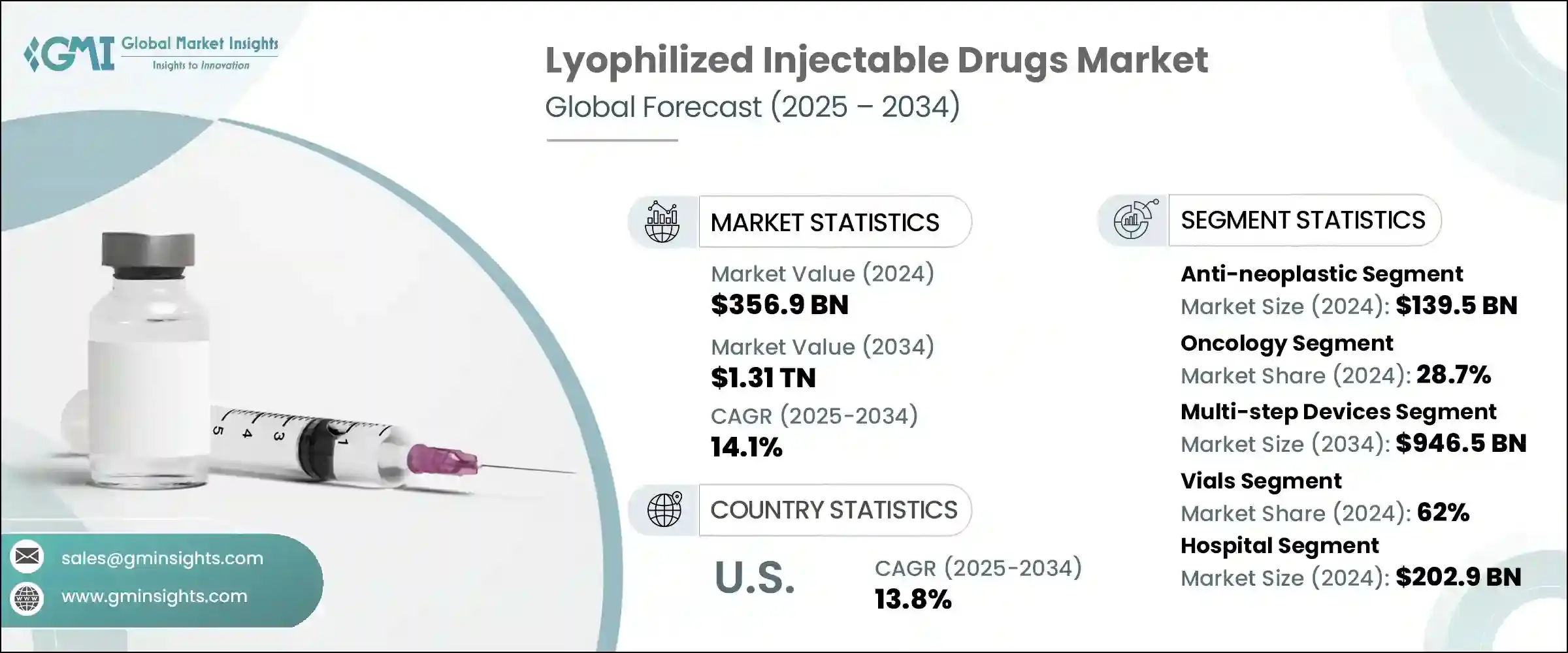
The global lyophilized injectable drugs market was estimated at USD 356.9 billion in 2024. The market is expected to grow from USD 400.8 billion in 2025 to USD 1.31 trillion in 2034, growing at a CAGR of 14.1%. The market's significant growth is driven by several factors, including the increasing burden of chronic diseases, rising demand for stable and effective biologics, advancements in freeze-drying technology, and expanding regulatory approvals.
Lyophilized Injectable Drugs Market Key Takeaways
Market Size & Growth
- 2024 Market Size: USD 356.9 Billion
- 2034 Forecast Market Size: USD 1.31 Trillion
- CAGR (2025–2034): 14.1%
Key Market Drivers
- Growing prevalence of chronic and infectious diseases.
- Technological advancements in drug delivery systems.
- Rising demand for biologics and complex molecules.
Challenges
- High production and equipment costs.
- Regulatory and quality compliance challenges.
Opportunity
- Personalized and precision medicine.
- Expansion of Contract Research and Manufacturing Services (CRAMS).
Get Market Insights & Growth Opportunities
The market is experiencing significant growth due to factors such as the rising prevalence of chronic diseases, increasing demand for stable and effective biologics, advancements in freeze-drying technology, and growing regulatory approvals. The surge in chronic conditions like cancer, infectious diseases, and autoimmune disorders has amplified the need for stable, long-shelf-life drug formulations, particularly biologics. This trend has established lyophilized formulations as a vital solution, driving the market's expansion.
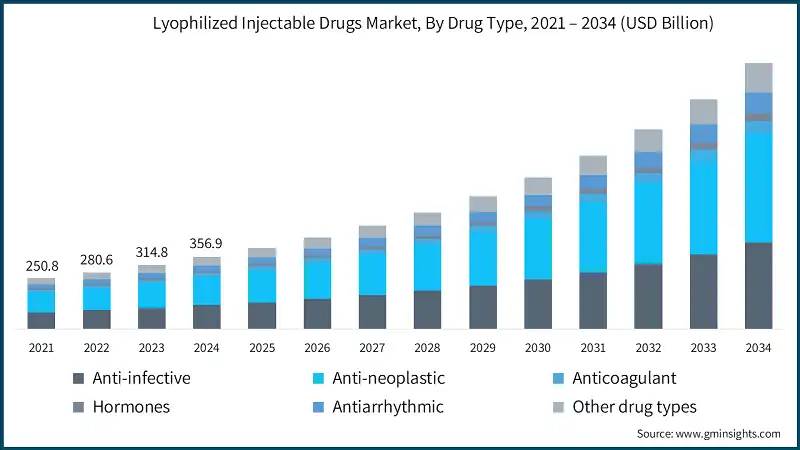
The market substantially increased from USD 250.8 billion in 2021 to USD 314.8 billion in 2023. The market growth is driven by the increasing prevalence of chronic and infectious diseases, rising adoption of biologics, and advancements in sterile manufacturing technologies. Expanding regulatory approvals have also significantly influenced market expansion. For example, according to the LyoHub 2023 annual report, submissions for lyophilized drug approvals increased by an average of 15% over the past decade. Between 2012 and 2022, regulatory agencies approved a total of 336 lyophilized drug products. In 2022 alone, the U.S. Food and Drug Administration (FDA) approved approximately 32 lyophilized drugs, with oncology and infectious disease treatments accounting for 82% of these approvals. This upward trend in approvals highlights the growing demand for lyophilized drug formulations, reinforcing their importance in the pharmaceutical industry and contributing to the market's projected growth.
Advancements in biologics and therapeutic injectable development, such as single-dose formulations, mRNA technology, scalable manufacturing processes, and improved cold-chain logistics, are expected to drive the growth of lyophilized injectables. These innovations enhance distribution efficiency and accessibility on a global scale. The increasing focus on improving injectable formulations is anticipated to boost the demand for lyophilized drugs, particularly in developing regions, thereby expanding the market's reach.
The lyophilized injectable drugs market is categorized by drug types, including anti-infective, anti-neoplastic, anticoagulant, hormones, antiarrhythmic, and others. These drugs require reconstitution with a solvent before administration. Lyophilization has emerged as the preferred method for developing heat- and moisture-sensitive compounds, particularly biologics and oncology drugs. Its ability to improve stability, extend shelf life, and maintain potency underscores its increasing adoption, presenting significant opportunities for market growth.
To get key market trends
Lyophilized Injectable Drugs Market Trends
- The market is expected to witness significant growth, driven by clinical advancements, technological innovations, and evolving regulatory frameworks.
- Increasing demand for biologics and complex molecules is fueling the need for lyophilization techniques, which enhance drug stability and extend shelf life.
- With biologics dominating pharmaceutical pipelines, particularly in oncology, autoimmune, and infectious diseases, the adoption of freeze-dried formulations is accelerating.
- Technological advancements in dual-chamber syringes, prefilled devices, and automated lyophilization systems are improving drug delivery efficiency and patient compliance, especially in home-based care and chronic disease management.
- Furthermore, pharmaceutical companies are increasingly outsourcing sterile injectable production to contract manufacturing organizations (CMOs) to achieve cost efficiency and scalability. Emerging markets, particularly in Asia-Pacific and Latin America, are experiencing rapid infrastructure development, creating new opportunities for market expansion.
Lyophilized Injectable Drugs Market Analysis
Learn more about the key segments shaping this market
The global market was valued at USD 250.8 billion in 2021. The market size reached USD 314.8 billion in 2023, from USD 280.6 billion in 2022.
Based on the drug type, the market is segmented into anti-infective, anti-neoplastic, anticoagulant, hormones, antiarrhythmic, and other drug types. The anti-neoplastic segment accounted for the largest revenue of USD 139.5 billion in 2024 due to the rising cancer burden globally with increasing use of biologics and cytotoxic agents in oncology treatment. The segment is expected to exceed USD 536 billion by 2034, growing at a CAGR of 14.6% during the forecast period.
- Lyophilization is particularly advantageous for anti-neoplastic drugs due to its key attributes, including enhanced stability, extended shelf life, and effectiveness for sensitive and high-value formulations.
- The preference for freeze-dried injectables in cancer care is further driven by the need for precise dosing, long-term storage, and reduced risk of contamination.
- Additionally, the increasing regulatory approvals for lyophilized drugs, coupled with advancements in drug formulation technologies addressing stability challenges, have intensified pharmaceutical companies' focus on developing advanced lyophilized products.
- For example, in June 2024, the U.S. FDA approved a new drug application (NDA) for Tepylute, a ready-to-dilute formulation for treating adenocarcinoma of the breast and ovary. Tepylute is a liquid form of the standard-of-care agent, thiotepa. The recommended dose of the agent is 0.3 mg/kg to 0.4 mg/kg, administered intravenously at 1 to 4-week intervals. This approval highlights significant progress in therapeutic options and formulations for cancer treatment.
- As oncology continues to dominate the global burden, the demand for lyophilized anti-neoplastic drugs is expected to remain strong, reinforcing this segment’s leadership in market revenue.
- Meanwhile, the antiarrhythmic segment is projected to witness significant growth, driven by the rising prevalence of cardiovascular disorders and the need for stable, rapid onset injectables in emergency care settings.
Learn more about the key segments shaping this market
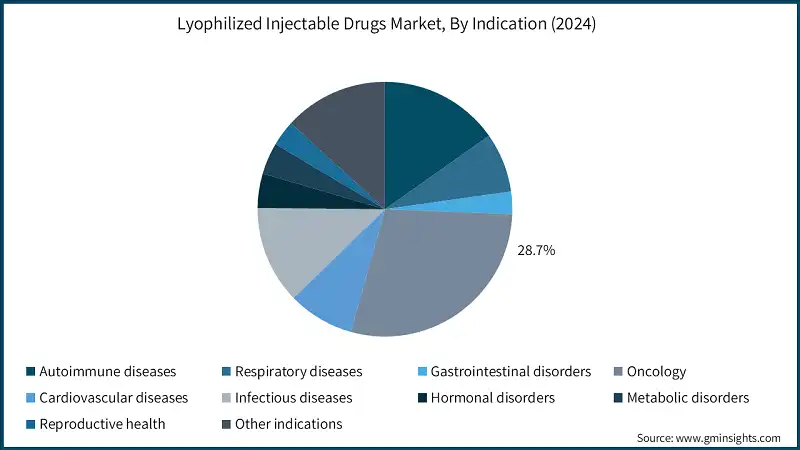
Based on the indication, the lyophilized injectable drugs market is segmented into autoimmune diseases, respiratory diseases, gastrointestinal disorders, oncology, cardiovascular diseases, infectious diseases, hormonal disorders, metabolic disorders, reproductive health, and other indications. The oncology segment accounted for the highest market share of 28.7% in 2024 due to the growing cancer burden.
- The oncology segment continues to dominate, driven by the rising prevalence of cancer and the demand for high-potency, stable formulations, such as chemotherapy agents.
- The growing need for formulation stability has led pharmaceutical companies to focus on cost-effective and reliable delivery systems to address the global healthcare burden.
- In February 2024, Fresenius Kabi launched Cyclophosphamide for Injection, a generic alternative to Cytoxan, for treating certain types of cancer in the U.S. This launch strengthens Fresenius Kabi’s extensive oncology portfolio and expands critical treatment options.
- Furthermore, the growth of targeted therapies and complex oncology treatment regimens underscores the increasing demand for freeze-dried injectables, further solidifying the oncology segment’s leadership in market revenue.
- Meanwhile, the gastrointestinal (GI) disorders segment is expected to witness robust growth in the market, driven by the rising incidence of GI conditions such as inflammatory bowel disease (IBD) and acute gastrointestinal bleeding.
Based on application, the lyophilized injectable drugs market is segmented into prefilled diluent syringes and multi-step devices. The multi-step devices segment accounted for the highest market share in 2024 and is expected to reach USD 946.5 billion by 2034 due to higher adoption of multi-step devices.
- Multi-step devices integrate both the solvent and lyophilized drug within a dual-chamber format.
- These devices operate by activating the central stopper to mix the components before administration, ensuring consistent dosing and minimizing contamination risks, making them ideal for high-potency or temperature-sensitive therapies.
- This advanced delivery mechanism supports complex biologics and oncology injectables that require precise reconstitution, gaining significant traction in the market.
- With the rising demand for self-use and cartridge-based formats that enhance dosing accuracy and patient safety, multi-step devices are increasingly preferred, reinforcing their dominance in the market.
- Meanwhile, prefilled diluent syringes are expected to witness robust growth in the market due to their ready-to-use (RTU) design, which improves dosing accuracy and reduces preparation errors. Their convenience and safety benefits are driving broader adoption in both clinical and self-administration settings.
Based on the packaging, the lyophilized injectable drugs market is segmented into vials, cartridges, and prefilled devices. The vials segment accounted for the highest market share of 62% in 2024.
- Vials are highly preferred due to their flexibility in accommodating variable dosages, ease of reconstitution, and ability to visually confirm the powder content.
- Moreover, vials offer superior sterility preservation until puncture and are robust enough for shipping and storing sensitive biologics, making them a preferred choice in healthcare settings.
- Vials support precise dosing and reduce contamination risks, making them particularly suitable for high-value, temperature-sensitive lyophilized formulations. Their compatibility with a wide range of drug formulations, especially biologics and oncology injectables, reinforces their market dominance.
- Additionally, ongoing innovations in vial design, such as ready-to-use (RTU) formats and enhanced glass materials, further strengthen their appeal among manufacturers and healthcare providers, securing their leadership in the packaging segment.
- On the other hand, prefilled devices are expected to witness significant growth, driven by the increasing demand for convenient and accurate dosing while minimizing contamination risks. Their user-friendly design improves patient compliance, making them a preferred choice in both clinical and homecare environments.
Based on the end use, the lyophilized injectable drugs market is segmented into hospitals, specialty clinics, and other end users. The hospital segment accounted for the largest revenue of USD 202.9 billion in 2024 due to the wide availability of comprehensive treatment care and access to skilled healthcare professionals.
- Hospitals remain the primary health facility due to their crucial role in managing complex treatments that require freeze-dried injectables such as oncology, metabolic, and infectious disease therapies.
- They offer advanced infrastructure, sterile administration environments, and highly trained medical staff, which are essential for handling high-potency and temperature-sensitive products.
- Moreover, hospitals remain indispensable due to their capacity to handle inpatient care, emergency dosing, and large-scale distribution making the hospitals segment the cornerstone of demand for lyophilized injectable products in the pharmaceutical market.
- On the other hand, specialty clinics are poised to witness significant growth in the market due to their expanding role in outpatient care, chronic disease management, and oncology treatments. Their focus on precision therapies and increasing adoption of advanced drug delivery systems drives this segment’s robust growth.

Looking for region specific data?
North America dominated the global lyophilized injectable drugs market with the highest market share of 47% in 2024.
- North America's dominance can be attributed to its well-established pharmaceutical industry, advanced healthcare infrastructure, and high prevalence of chronic diseases in the region.
- The region benefits from robust regulatory support, significant R&D investments, and a substantial number of FDA approvals for lyophilized products, particularly in oncology, infectious diseases, and autoimmune therapies.
- Moreover, the increasing preference for convenient, long-shelf-life formulations in hospital and outpatient settings further drives market growth.
In 2024, the U.S. lyophilized injectable drugs market accounted for USD 151.8 billion revenue from USD 134.2 billion in 2023 and is anticipated to grow at a CAGR of 13.8% between the 2025 to 2034 period.
- The dominance of the U.S. market is attributed to its advanced pharmaceutical manufacturing capabilities, strong regulatory framework, and high demand for stable, long-acting injectable formulations.
- The country’s robust R&D infrastructure has facilitated the development of complex biologics and oncology therapeutics, which rely on lyophilization for stability and efficacy, reinforcing its leadership in the industry.
- Rising FDA approvals for lyophilized products and the growing adoption of ready-to-use (RTU) formats in hospitals and specialty clinics have further driven the demand for stable lyophilized injectables.
- Moreover, the presence of key pharmaceutical companies such as Pfizer, Merck, and Bristol Myers Squibb continues to support innovation and large-scale production, strengthening the U.S. market's position in this segment.
Germany is poised to achieve significant growth in the Europe lyophilized injectable drugs market.
- Germany is expected to witness significant growth due to its advanced healthcare infrastructure and favorable reimbursement policies, which are likely to drive the adoption of injectables, including lyophilized formulations.
- Additionally, the rising prevalence of cancer, infectious diseases, autoimmune disorders, and metabolic conditions is boosting the demand for stabilized biologics and antibiotic therapies.
- For example, according to data from the International Agency for Research on Cancer (IARC), approximately 605,805 cancer cases were diagnosed in Germany in 2022. This number is projected to increase to 745,209 by 2045. This growing cancer burden highlights the need for effective, long-shelf-life therapeutic solutions. As the demand for reliable oncology treatments rises, lyophilized injectable formulations are gaining prominence.
- Furthermore, Germany’s strong pharmaceutical ecosystem, supported by regulatory frameworks from the European Medicines Agency (EMA), advanced manufacturing capabilities, and high contract manufacturing activity, is driving market growth.
- As a result, Germany's robust pharmaceutical innovation and increasing demand for therapeutic treatments position it as a key market for lyophilized injectables in Europe.
Asia Pacific is poised to grow at a significant growth rate in the global lyophilized injectable drugs market over the forecast period.
- The Asia-Pacific region is expected to witness significant growth, driven by increasing healthcare expenditure, an expanding pharmaceutical manufacturing sector, and a rising prevalence of chronic diseases, which is boosting the demand for biologics and advanced treatment modalities.
- Countries such as China, India, Japan, and South Korea have large patient populations and a high prevalence of chronic and infectious diseases, creating substantial demand for long-acting, stable injectable therapies.
- Additionally, governments across the region are supporting advancements in healthcare infrastructure, implementing favorable regulations for biosimilars and generics, and investing in public health programs to raise awareness of advanced injectable therapies.
- The region also benefits from the presence of cost-effective contract manufacturing organizations (CMOs), making it an attractive hub for outsourcing lyophilized drug production.
- Furthermore, supportive regulatory reforms and strategic investments in sterile manufacturing infrastructure are driving market growth. As a result, the Asia-Pacific region is anticipated to emerge as one of the fastest-growing markets in the global market.
China is anticipated to grow significantly within the Asia Pacific lyophilized injectable drugs market.
- China's dominance in the market is driven by its large patient population, expanding pharmaceutical manufacturing capabilities, significant investments in healthcare infrastructure, and increasing demand for biologics and injectable therapies.
- For example, according to Global Cancer Observatory (GLOBOCAN) estimates, approximately 4 million new cancer cases were diagnosed in China in 2022, with lung cancer accounting for nearly 1 million cases. This substantial cancer burden highlights the critical need for advanced treatment modalities, which is expected to drive the adoption of lyophilized injectable drugs.
- Furthermore, China's growing role in global contract manufacturing and its advancements in drug delivery systems, such as prefilled syringes and dual-chamber cartridges, enhance its competitiveness in the market.
- Additionally, the country's strong focus on improving pharmaceutical infrastructure, bolstering R&D capabilities, and addressing chronic and infectious disease concerns, with an emphasis on drug stability and shelf-life enhancement, is accelerating the adoption of lyophilization technologies.
- These factors collectively position China as a key contributor to the growth of the Asia Pacific market in the coming years.
Brazil is projected to witness significant growth in Latin America lyophilized injectable drugs market in coming years.
- Brazil's robust growth is driven by its increasing elderly population, which is highly vulnerable to chronic diseases, thereby boosting the demand for stable, temperature-sensitive medications.
- The country’s strategic initiatives to improve access to advanced therapies, along with government efforts to support domestic production, are accelerating the adoption of lyophilized injectables.
- Furthermore, growing collaborations with global pharmaceutical companies and contract manufacturing organizations (CMOs) are enhancing Brazil’s capabilities in sterile injectable production, positioning it as a key growth hub in the region.
- These factors collectively position Brazil as a significant driver of growth in Latin America’s market in the coming years.
Saudi Arabia is anticipated to grow in the Middle East and African lyophilized injectable drugs market.
- Saudi Arabia's potential growth is driven by factors such as expanding pharmaceutical infrastructure, strategic investments in healthcare and innovation, and government initiatives promoting preventive healthcare measures.
- For instance, the government's Vision 2030 initiative, aimed at modernizing the healthcare sector, is fostering demand for high-quality injectable formulations, particularly in areas like oncology, infectious diseases, and autoimmune conditions. The Ministry of Health's expanded immunization strategy, which includes comprehensive protocols and broader healthcare coverage, is gaining significant traction in the country.
- The presence of major market players, along with the growing adoption of advanced drug delivery technologies such as lyophilization, is enhancing drug stability and shelf life in Saudi Arabia.
- Furthermore, regulatory reforms and public-private partnerships are accelerating clinical development and manufacturing capabilities.
- These factors collectively position Saudi Arabia as a key growth driver in the Middle East and Africa market.
Lyophilized Injectable Drugs Market Share
- Leading companies such as Sanofi, Merck, F. Hoffmann-La Roche, Pfizer, and Fresenius collectively hold approximately 40% of the market share. These companies aim to maintain leadership through their extensive portfolios, strong R&D capabilities, strategic partnerships, and global distribution networks.
- These firms are utilizing advanced lyophilization technologies to enhance product shelf life, improve drug stability, and ensure reliable drug delivery, with a significant focus on oncology, biologics, and vaccines.
- Pfizer and Merck dominate the market by continuously innovating and investing in novel lyophilized biologics and vaccines. Meanwhile, Sanofi and Roche maintain a strong market presence through their diversified therapeutic offerings and global production facilities.
- Additionally, Takeda Pharmaceuticals and Johnson & Johnson are concentrating on specialty injectables and oncology, leveraging robust pipelines and advanced freeze-drying processes to strengthen their competitive positions in the market.
- Emerging players such as Zydus, Cipla, and Aurobindo are gaining momentum through niche innovations, regulatory approvals, and product launches. Furthermore, advancements in technology, patent expirations, and the growing demand for temperature-sensitive biologics are intensifying market competition. Companies are increasingly adopting strategies such as strategic collaborations, acquisitions, and capacity expansions to secure market share and effectively address global healthcare demands.
Lyophilized Injectable Drugs Market Companies
Few of the prominent players operating in the lyophilized injectable drugs industry include:
- Akums Drugs and Pharmaceuticals
- Aurobindo Pharma
- Bora Pharmaceuticals
- Bristol Myers Squibb
- Cipla
- F. Hoffmann-La Roche
- Fareva
- Fresenius
- Gilead Sciences
- Gufic Group
- Johnson & Johnson
- Meiji Group
- Merck
- Novo Nordisk
- Pfizer
- Sanofi
- Takeda Pharmaceuticals
- Vetter Pharma
- Zydus
- Pfizer
Pfizer boasts a robust portfolio of high-stability lyophilized injectables across various therapeutic indications. The company employs advanced freeze-drying technologies and state-of-the-art sterile manufacturing facilities to ensure consistent drug quality, extended shelf life, and rapid reconstitution, catering to global hospital and critical care markets.
- F. Hoffmann-La Roche
Roche capitalizes on its expertise in oncology and biologics to deliver reliable lyophilized formulations that ensure therapeutic stability and patient safety. Its integrated R&D capabilities and proprietary drug delivery platforms facilitate the development of temperature-sensitive monoclonal antibodies and diagnostics, enhancing treatment precision and efficacy.
- Sanofi
Sanofi’s leadership in vaccines and biologics extends to lyophilized injectables, offering ease of transportation and storage. The company leverages advanced R&D capabilities, manufacturing expertise, and cold-chain distribution services to deliver stable, ready-to-use formulations for various disease indications. Additionally, its global presence and focus on underserved regions strengthen its position in the lyophilized injectable drugs market.
- Merck
Merck maintains its leadership through advanced lyophilization facilities that enable the consistent, high-quality production of stable lyophilized injections and biologics. The company’s strong regulatory approvals, significant R&D investments, and public-private collaborations support its ability to meet the growing demand for cutting-edge lyophilized formulations, reinforcing its market leadership and innovation.
Lyophilized Injectable Drugs Market Report Attributes
| Key Takeaway | Details |
|---|---|
| Market Size & Growth | |
| Base Year | 2024 |
| Market Size in 2024 | USD 356.9 Billion |
| Forecast Period 2025 - 2034 CAGR | 14.1% |
| Market Size in 2034 | USD 1.31 Trillion |
| Key Market Trends | |
| Drivers | Impact |
| Growing prevalence of chronic and infectious diseases | • The rising prevalence of cancer, autoimmune disorders, and infectious diseases is driving the demand for injectable therapies, increasing the need for lyophilization to ensure long-term storage and stability. |
| Technological advancements in drug delivery systems | • Technological advancements, such as dual-chamber syringes and prefilled cartridges, are enhancing patient compliance and improving the efficiency of drug administration. |
| Rising demand for biologics and complex molecules | • The growing adoption of biologics, peptides, and monoclonal antibodies in therapeutic applications underscores the importance of stable formulations, making lyophilization critical for preserving drug efficacy. |
| Pitfalls & Challenges | Impact |
| High production and equipment costs | • Lyophilization demands specialized equipment and expertise, resulting in significant capital and operational expenses. |
| Regulatory and quality compliance challenges | • Stringent regulatory requirements for sterility, stability, and GMP compliance present challenges, particularly for smaller manufacturers. |
| Opportunities: | Impact |
| Personalized and precision medicine | • The growing focus on targeted therapies and personalized medicine is driving the demand for customized lyophilized formulations designed to meet individual patient requirements. |
| Expansion of Contract Research and Manufacturing Services (CRAMS) | • Pharmaceutical companies are increasingly outsourcing lyophilization processes to CRAMS to optimize production scalability and reduce operational costs. |
| Market Leaders (2024) | |
| Market Leader |
|
| Top Players |
Collective market share in 2024 is 40% |
| Competitive Edge |
|
| Regional Insights | |
| Largest Market | North America |
| Fastest Growing Market | Asia Pacific |
| Emerging Countries | India, Brazil, South Korea, and South Africa |
| Future outlook |
|
What are the growth opportunities in this market?
Lyophilized Injectable Drugs Industry News:
- In June 2025, Fareva expanded its new Good Manufacturing Practices (GMP) pilot unit in Pau, enhancing its capabilities in the development and production of liquid and lyophilized injectable formulations. This strategic expansion strengthens the company’s service offerings in formulation development and aseptic filling, specifically targeting early-phase clinical trial batch production and small-scale commercial manufacturing. This move reinforces Fareva’s manufacturing and production capabilities in the market.
- In January 2025, Akums Drugs and Pharmaceuticals expanded its manufacturing facility to focus on lyophilized products. The facility is equipped to manufacture vials, form-fill-seal (FFS) products, and eye and ear drops. This strategic expansion aims to enhance the company’s sterile dosage form capabilities.
- In August 2024, Bora Pharmaceuticals acquired Emergent BioSolutions’ sterile manufacturing facility in Baltimore-Camden, Maryland, adding drug product fill/finish capabilities to its biologics drug substance portfolio. This acquisition enables Bora Pharmaceuticals to offer clinical and commercial non-viral aseptic fill/finish services for vials and pre-filled syringes across four fill lines, along with lyophilization, formulation development, and support services. The acquisition enhances Bora’s capabilities in sterile formulations, expanding its drug product manufacturing services across the U.S.
The lyophilized injectable drugs market research report includes an in-depth coverage of the industry with estimates and forecast in terms of revenue in USD Million and from 2021 - 2034 for the following segments:
Market, By Drug Type
- Anti-infective
- Anti-neoplastic
- Anticoagulant
- Hormones
- Antiarrhythmic
- Proton pump inhibitors
- Anesthetics
- Other drug types
Market, By Indication
- Autoimmune diseases
- Respiratory diseases
- Gastrointestinal disorders
- Oncology
- Cardiovascular diseases
- Infectious diseases
- Hormonal disorders
- Metabolic disorders
- Reproductive health
- Other indications
Market, By Application
- Prefilled diluent syringes
- Multi-step devices
Market, By Packaging
- Vials
- Cartridges
- Prefilled devices
Market, By End Use
- Hospitals
- Specialty clinics
- Other end use
The above information is provided for the following regions and countries:
- North America
- U.S.
- Canada
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Netherlands
- Asia Pacific
- China
- Japan
- India
- Australia
- South Korea
- Latin America
- Brazil
- Mexico
- Argentina
- Middle East and Africa
- South Africa
- Saudi Arabia
- UAE
Frequently Asked Question(FAQ) :
What is the market size of the lyophilized injectable drugs in 2024?
The market size was USD 356.9 billion in 2024, with a CAGR of 14.1% expected through 2034, driven by the increasing prevalence of chronic diseases and advancements in freeze-drying technology.
What is the projected value of the lyophilized injectable drugs market by 2034?
The market is anticipated to reach USD 1.31 trillion by 2034, owing to the rising demand for stable biologics and expanding regulatory approvals.
What is the projected size of the lyophilized injectable drugs market in 2025?
The market is expected to grow to USD 400.8 billion in 2025, supported by technological innovations and the growing adoption of freeze-dried formulations.
How much revenue did the oncology segment generate?
The oncology segment accounted for the highest market share of 28.7% in 2024.
What was the valuation of the multi-step devices segment?
The multi-step devices segment accounted for the highest market share in 2024 and is projected to reach USD 946.5 billion by 2034.
Which region leads the lyophilized injectable drugs market?
The U.S. led the market with USD 151.8 billion in revenue in 2024, supported by advanced pharmaceutical manufacturing capabilities and a strong regulatory framework.
What are the upcoming trends in the lyophilized injectable drugs industry?
Key trends include the increasing adoption of lyophilization techniques for biologics, advancements in freeze-drying technology, and the growing focus on stable, long-acting injectable formulations.
Who are the key players in the lyophilized injectable drugs market?
Key players include Akums Drugs and Pharmaceuticals, Aurobindo Pharma, Bora Pharmaceuticals, Bristol Myers Squibb, Cipla, F. Hoffmann-La Roche, Fareva, and Fresenius.
Lyophilized Injectable Drugs Market Scope
Related Reports