Summary
Table of Content

In-Vitro Diagnostics Packaging Market
Get a free sample of this report
Form submitted successfully!
Error submitting form. Please try again.
Thank you!
Your inquiry has been received. Our team will reach out to you with the required details via email. To ensure that you don't miss their response, kindly remember to check your spam folder as well!

Request Sectional Data
Thank you!
Your inquiry has been received. Our team will reach out to you with the required details via email. To ensure that you don't miss their response, kindly remember to check your spam folder as well!
Form submitted successfully!
Error submitting form. Please try again.
In-Vitro Diagnostics Packaging Market Size
The growth of the in-vitro diagnostics packaging market is attributed to rising volume of infectious disease diagnostics, growth of point-of-care testing, increasing regulatory demands for sterile packaging, growth of home-based diagnostics, and the rising demand for cold chain solutions.
In-Vitro Diagnostics Packaging Market Key Takeaways
Market Size & Growth
- 2025 Market Size: USD 8.9 Billion
- 2026 Market Size: USD 9.2 Billion
- 2035 Forecast Market Size: USD 14.9 Billion
- CAGR (2026–2035): 5.5%
Regional Dominance
- Largest Market: North America
- Fastest Growing Region: Asia Pacific
Key Market Drivers
- Rising infectious disease diagnostics volume globally.
- Growth in point-of-care testing kits demand.
- Cold chain logistics expansion for temperature-sensitive IVDs.
- Stringent regulatory requirements for sterile packaging.
- Increasing home-based diagnostic testing adoption.
Challenges
- Supply chain disruptions in medical-grade plastics.
- Complex validation requirements for sterile packaging.
Opportunity
- Sustainable and recyclable medical packaging innovations.
- Smart packaging integration for temperature monitoring.
Key Players
- Market Leader: Amcor led with over 16.5% market share in 2025.
- Leading Players: Top 5 players in this market include Amcor, Aptargroup, Corning, Greiner Holding, Oliver, which collectively held a market share of 55.2% in 2025.
Get Market Insights & Growth Opportunities
The IVD packaging market is driven by the rising global volume of infectious disease diagnostics. The rising incidence rate of infectious diseases such as COVID-19 and tuberculosis continues to fuel the need for diagnostic tests. According to the World Health Organization's Global Tuberculosis Report 2025, in 2024, there were 10.7 million people who fell ill due to TB in the world. This rising need for testing further increases the need for sterile single-use diagnostic packaging, thus emphasizing the importance of diagnostic packaging in ensuring safe large-scale testing and disease detection.
Additionally, the growth of the IVD packaging market is supported by increasing demand for point-of-care (POC) testing kits. The move towards decentralized testing is driving demand for portable, user-friendly packaging. In 2025, the United States Food and Drug Administration approved additional at-home and rapid diagnostic tests through its expanded emergency use and clearance program. This further increases the need for secure and tamper-evident packaging, emphasizing its importance in relation to safe handling and usability outside the laboratory.
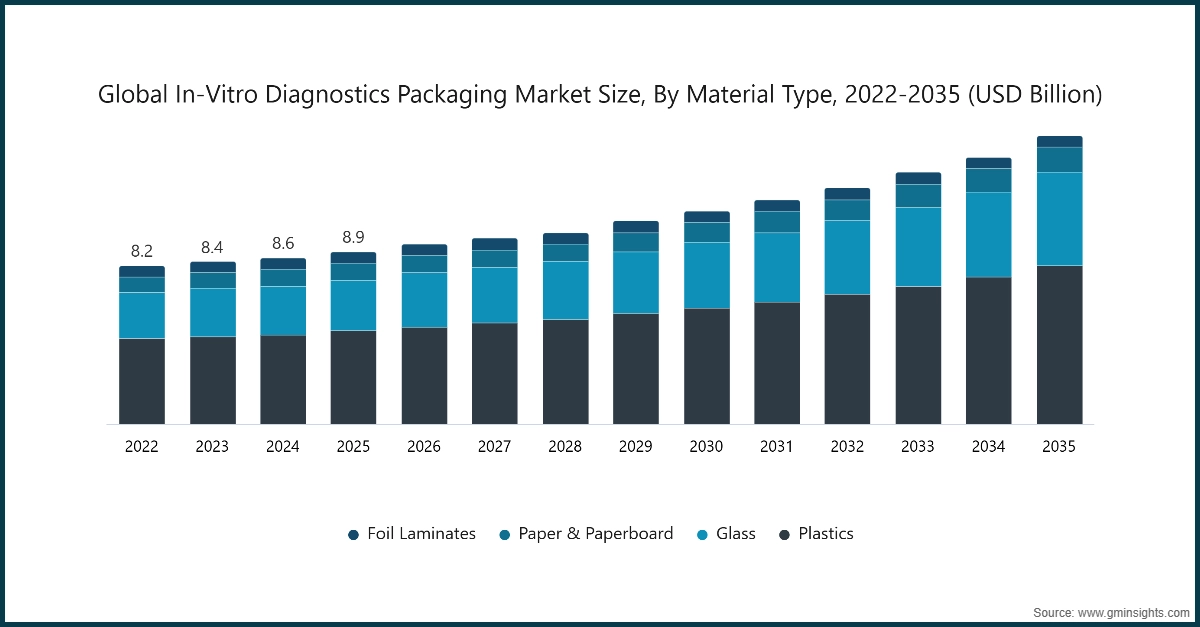
The in-vitro diagnostics packaging market increased steadily from USD 8.2 billion in 2022 and reached USD 8.6 billion in 2024. The period experienced growth owing to increased diagnostic testing because of infectious disease outbreaks, the development of rapid point-of-care kits and the implementation of international standards for sterile packaging. The market experienced additional growth because healthcare organizations adopted cold chain logistics to handle temperature-sensitive IVD products, home-testing programs increased and financial resources were directed toward modernizing healthcare supply chains.
The global in-vitro diagnostics packaging market was valued at USD 8.9 billion in 2025. The market is expected to grow from USD 9.2 billion in 2026 to USD 11.6 billion in 2031 & USD 14.9 billion in 2035, at a CAGR of 5.5% during the forecast period according to the latest report published by Global Market Insights Inc.
To get key market trends
In-Vitro Diagnostics Packaging Market Trends
- The shift towards more sustainable and less carbon-intensive packaging materials is emerging as another important trend for IVD packaging. The trend started in 2021 when healthcare systems and regulators began their push to decrease medical waste and carbon emissions. The trend will persist until 2032 because organizations will implement stricter environmental regulations and corporate sustainability initiatives. The current market trend forces manufacturers to develop packaging solutions which include recyclable materials and bio-based components and single-material structures.
- Adoption of automation and high-speed packaging solutions is a new trend that is changing IVD packaging. This trend is gaining momentum, especially after 2020, with a focus on increasing demand for IVD diagnostics and error-free production processes. This trend is expected to grow up to 2030, especially with a focus on increasing production, minimizing errors, and enhancing quality, especially in sterile packaging processes.
- The increase in customization and modular packaging formats is also emerging as a promising trend in the IVD industry. This trend has been emerging since 2022, as there has been an increase in the diversity of product portfolios in the diagnostic industry, which has led to an increase in flexible packaging solutions. The trend will continue through 2031 to support evolving diagnostic formats and personalized healthcare needs. The system multiplies product launch speed while achieving optimal test equipment performance and user-friendly operation.
- The trend of increasing adoption of advanced barrier technology in packaging is also affecting product safety and performance. This trend has been gaining momentum since 2021, as there has been an increasing need to extend product life and protect sensitive reagents in the diagnostic industry. The trend will continue until 2030 because diagnostic tests are becoming more intricate. The results create enhanced product stability which decreases contamination risks and improves supply chain reliability for international operations.
In-Vitro Diagnostics Packaging Market Analysis
Learn more about the key segments shaping this market
Based on material type, the in-vitro diagnostics packaging market is divided into foil laminates, paper & paperboard, glass, and plastics.
- The plastics segment led the market in 2025, holding a 54% share. Plastic dominates the IVD packaging market, because they offer multiple uses, affordable pricing and can be used with single-use diagnostic products which include tubes, vials and microplates. The materials provide essential features including durability, chemical resistance and sterilization compatibility for laboratory and hospital environments which require clean handling of materials and production at high volumes.
- The glass segment is anticipated to grow at a CAGR of 6.2% over the forecast period. This growth is driven by increasing demand for high-purity, chemically inert, and thermally stable packaging materials, especially for sensitive reagents and biological materials. Glass vials, ampoules, and sample containers are preferred for high-end, specialized diagnostic procedures where minimal interaction between materials is required, thus facilitating pharmaceutical diagnostics and high-end laboratory testing.
Based on packaging product type, the in-vitro diagnostics packaging market is divided into bottles & vials, tubes & microtubes, pouches & bags, trays & blisters, test cartridges & cassettes, cartons & boxes, and others.
- The bottles & vials segment dominated the market in 2025, valued at USD 2.8 billion, due to its extensive use for storing diagnostic reagents, culture media, and samples. Their compatibility with automated dispensing systems, sterilization processes, and cold chain requirements makes them a preferred format in hospitals, laboratories, and centralized testing facilities. High chemical resistance, secure sealing, and consistent sizing ensure regulatory compliance and reliable diagnostic performance.
- The tubes & microtubes segment is expected to witness the growth at a CAGR of 5.9% over the forecast period. This growth is driven by the rising adoption of high-throughput testing, molecular diagnostics, and point-of-care kits that require compact, leak-proof, and precise sample containers. Their adaptability for centrifugation, automated processing, and home-testing applications accelerates demand across decentralized and laboratory testing workflows.
Learn more about the key segments shaping this market
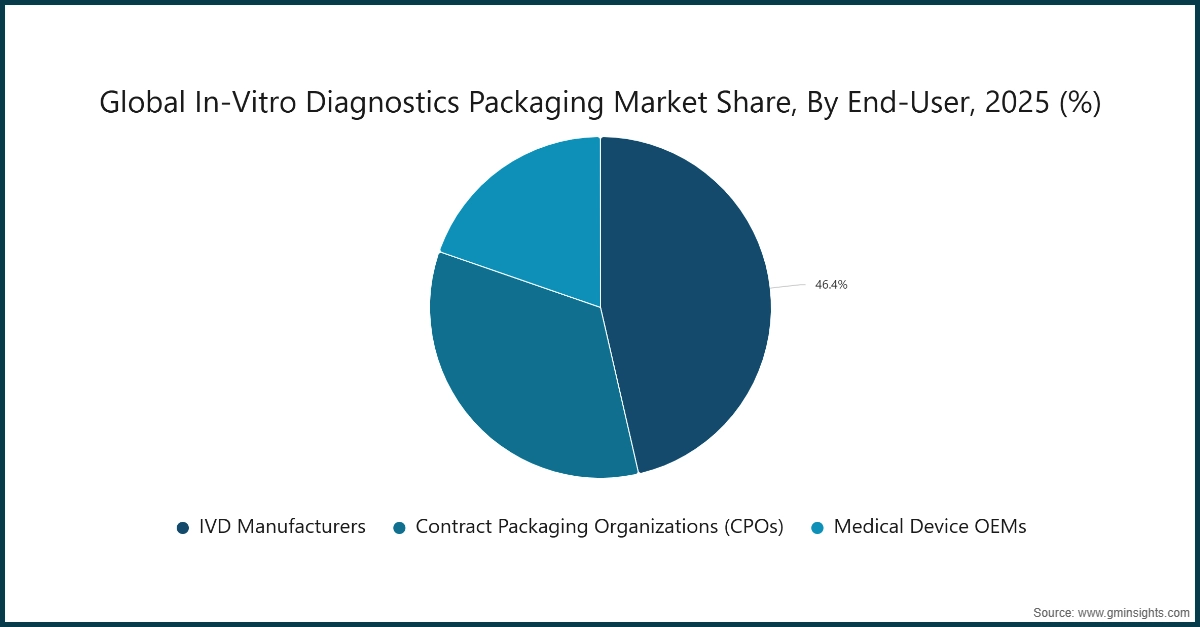
Based on end-user, the in-vitro diagnostics packaging market is divided into IVD manufacturers, contract packaging organizations (CPOs), and medical device OEMs.
- The IVD manufacturers segment led the market in 2025 with a market share of 46.4%. These manufacturers are the largest consumers of diagnostic packaging, as they require sterile, high-quality, and regulatory-compliant containers for reagents, kits, and consumables. Large-scale production, consistent quality control, and integration with automated laboratory systems make IVD manufacturers the dominant end-user segment, ensuring continuous demand for advanced packaging solutions.
- The contract packaging organizations (CPOs) segment is expected to grow at a CAGR of 7% during the forecast period. Growth is driven by outsourcing trends, where diagnostic companies rely on CPOs for specialized sterile packaging, flexible batch production, and cold chain compliance. The scalability, customization, and regulatory expertise offered by CPOs make them increasingly preferred partners for emerging diagnostics and rapid product launches.
Looking for region specific data?
North America In-Vitro Diagnostics Packaging Market
North America held a share of 35.1% of IVD packaging market in 2025.
In North America, the global in-vitro diagnostics packaging industry is expanding due to stringent regulatory requirements around sterility, contamination control, and cold chain management in diagnostics. Increasing demand for rapid and point-of-care testing kits, especially in healthcare and research sectors, drives adoption of innovative packaging solutions.
- Major investments by healthcare providers and diagnostic manufacturers in scalable, automated, and eco-friendly packaging technologies support sustained growth. The region leads in integrating smart packaging features for temperature monitoring and supply chain traceability, with steady expansion expected through 2035.
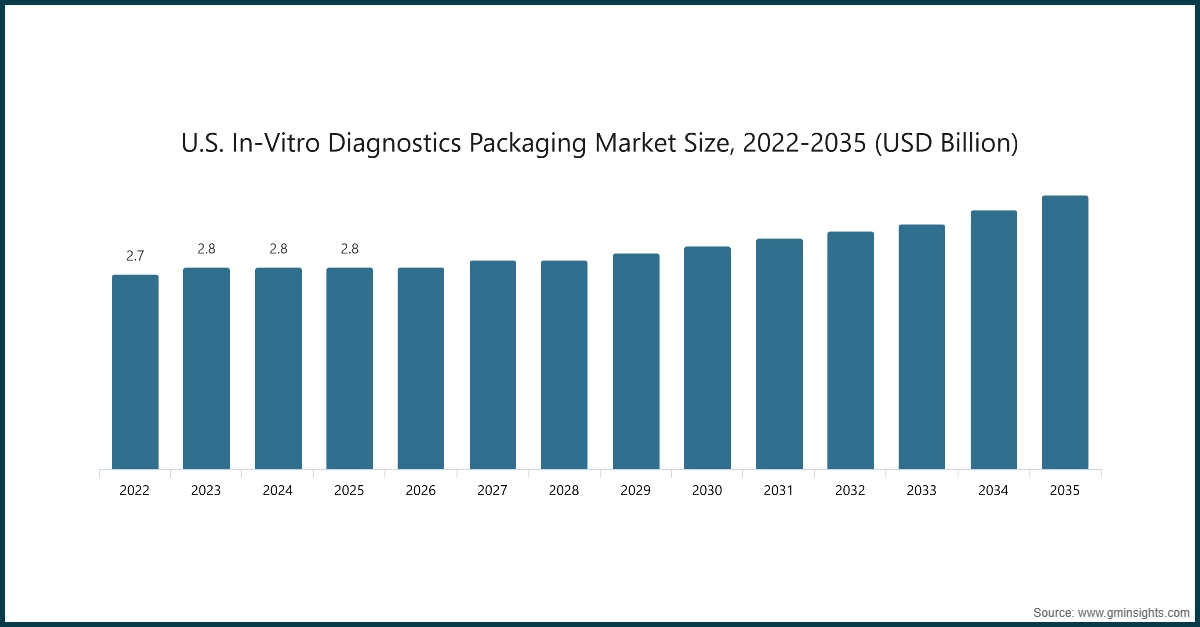
The U.S. in-vitro diagnostics packaging market was valued at USD 2.7 billion and USD 2.8 billion in 2022 and 2023, respectively. The market size reached USD 2.8 billion in 2025, growing from USD 2.8 billion in 2024.
- The growth of the market for IVD packaging in the U.S. is particularly strong because of targeted federal funding to expand diagnostic testing and laboratory capacity. In January 2025, the CDC announced NOFO CD‑25‑0019, a multi-year funding initiative through 2030 to strengthen public health laboratories for infectious and emerging disease testing, improve workforce training, and expand high-volume diagnostic capabilities.
- Along with updated FDA and CDC requirements for sterile, tamper-evident, and cold-chain compliant packaging, these investments are driving adoption of advanced IVD packaging formats, insulated vials, and barrier containers, making the U.S. the leading market within North America for safe, scalable, and regulatory-compliant diagnostic packaging solutions.
Europe In-Vitro Diagnostics Packaging Market
Europe market accounted for USD 2.5 billion in 2025 and is anticipated to show lucrative growth over the forecast period.
- The IVD packaging market in Europe is expanding due to increasing regulations on medical device safety and traceability under the EU Medical Device Regulation (MDR) 2017/745, which enforces strict sterilization, labeling, and quality standards for diagnostic consumables and packaging. Compliance with MDR has driven demand for validated sterile barrier systems, traceable tamper‑evident formats, and validated packaging processes across diagnostics supply chains.
- Countries such as Germany, France, and the Netherlands are investing in national diagnostic infrastructure upgrades, including EU‑backed public health lab modernization and advanced cold chain logistics frameworks. These investments ensure sustained adoption of high‑integrity packaging formats that support compliance, improved diagnostic accuracy, and decentralized point‑of‑care testing across the region.
Germany dominates the Europe in-vitro diagnostics packaging industry, showcasing strong growth potential.
- Germany leads Europe’s IVD packaging market, driven by high healthcare investment and stringent regulatory compliance. Total healthcare expenditure reached 11.7% of GDP in 2023, the highest in the EU, supporting expansion of diagnostic testing infrastructure and demand for high‑integrity, sterile packaging solutions. Strict enforcement of the EU In Vitro Diagnostic Medical Devices Regulation (IVDR) and domestic requirements mandates sterility, labeling, and traceability, prompting manufacturers to adopt advanced barrier packaging, cold chain–ready vials, and tamper‑evident formats to meet both domestic and export standards.
- Furthermore, the phased implementation of Eudamed database modules beginning in May 2026 will require unique device identification and comprehensive documentation for diagnostics and associated packaging, reinforcing Germany’s position as an early adopter of strengthened regulatory traceability and quality‑assured packaging systems.
Asia Pacific In-Vitro Diagnostics Packaging Market
The Asia Pacific market is anticipated to grow at the highest CAGR of 8.6% during the forecast period.
- The Asia Pacific IVD packaging market is expanding rapidly because healthcare infrastructure investments are increasing, diagnostic testing volumes are rising and governments are launching programs to improve public health systems. Countries are investing public funds to improve their laboratory networks and diagnostic supply chains which drives the need for packaging solutions that maintain sterile conditions and prevent unauthorized access while supporting cold chain storage requirements.
- The ASEAN Medical Device Directive updates (2025) establish regulatory harmonization efforts which create standardized packaging and labeling requirements throughout the region thereby increasing the use of validated high‑integrity formats. The expansion of private sector businesses by both multinational and local companies drives production capacity growth while enabling technology transfers that will help advanced IVD packaging technologies spread throughout the region until the end of the forecast period.
China in-vitro diagnostics packaging market is estimated to grow with a significant CAGR, in the Asia Pacific market.
- China demonstrates rapid development of IVD packaging because its Healthy China 2030 initiative seeks to enhance diagnostic testing access while building up domestic production capabilities. The National Medical Products Administration (NMPA) has tightened regulatory requirements for packaging sterility, labeling, and traceability for in‑vitro diagnostics, which forces manufacturers to adopt advanced barrier materials and serialization technologies.
- The expansion of county-level CDC lab networks and major investments in TB, hepatitis, and cancer screening programs across China create a greater need for standardized packaging solutions that meet high-quality standards. The growth of local contract manufacturing and outsourcing partners in China enables better access to customized packaging formats, which support the country's various diagnostic needs.
Middle East and Africa In-Vitro Diagnostics Packaging Market
UAE market to experience substantial growth in the Middle East and Africa.
- The IVD packaging market in the United Arab Emirates (UAE) experiences substantial expansion because of the country's growing healthcare facilities and its government-backed diagnostic programs. The Health Strategy of the UAE Ministry of Health and Prevention for the period 2025 to 2031 aims to build laboratory networks and emergency response systems while acquiring sterile diagnostic supplies and secure packaging materials.
- The construction of modern cold chain logistics facilities at Dubai and Abu Dhabi International Airports has enhanced the temperature-controlled processing system for incoming reagents and biologics, which has created an increased need for insulated vials, barrier pouches and temperature-controlled packaging products.
- The UAE National Genome Strategy uses public-private partnerships to expand precision diagnostics and genomic testing services which need certified tamper-evident containers and labeling that meets both GCC and international standards. These developments have placed the UAE in a strategic position to become a hub for high-quality IVD packaging production and distribution in the Middle East and Africa regions due to a high volume of medical tourism and export activities from the UAE.
In-Vitro Diagnostics Packaging Market Share
The IVD packaging market is led by players such as Amcor, AptarGroup, Corning Incorporated, Greiner Holding AG, and Oliver Healthcare Packaging. These companies collectively held a market share of 55.2% in 2025. These companies have a competitive advantage due to their diversified product offerings, such as plastics, glass, and high-end sterile packaging materials, strong compliance, and global manufacturing and distribution networks.
Investment in R&D, green materials, smart packaging solutions, and IVD manufacturers' partnerships enables these companies to increase their market share considerably in geographical regions such as North America, Europe, and Asia-Pacific. These companies have also focused their efforts on smart integration, such as cold chain solutions, enabling real-time monitoring, traceability, etc., thus strengthening their market leadership even further.
In-Vitro Diagnostics Packaging Market Companies
Prominent players operating in the in-vitro diagnostics packaging industry are as mentioned below:
- Abdos Labtech Private Limited
- ACADEMY MEDICAL INSTRUMENTS Limited Company
- Ajosha Bio Teknik Pvt. Ltd.
- AdvaCare Pharma
- Amcor Limited
- Amrit Scientific Ind. Private Limited
- Aptargroup Incorporated
- Corning Incorporated
- Focus Technology Co., Ltd.
- FUSION BIOTECH
- Globe Scientific Inc.
- Greiner Holding AG
- Kindly (KDL) Group
- Labtech Disposables
- Narang Medical Limited
- Oliver
- Peacock Industries
- VWR International, LLC.
- YuHuan KangJia
- Zhuhai Ideal Biotech Co., Ltd
Amcor develops sustainable packaging solutions which maintain high barrier protection while providing sterile packaging for IVD consumables. The company uses advanced plastics and multilayer films with bio-based materials to create containers and vials and pouches which prevent contamination. The company's worldwide manufacturing network together with its regulatory knowledge and research development commitments allows it to produce scalable solutions for laboratories, hospitals and point-of-care settings.
AptarGroup offers precision dispensing, sealing, and closure solutions for diagnostic kits. AptarGroup offers a wide range of uncontaminated packaging solutions that include tamper-evident, leak-resistant, and user-friendly packaging solutions for centralized lab testing kits as well as home testing kits. Innovation is a key factor that has helped AptarGroup maintain a strong presence in the global market.
Corning Incorporated delivers chemically resistant, thermally stable glass vials, ampoules, and containers for sensitive reagents and biologics. Its expertise in high-purity glass manufacturing and global regulatory compliance ensures long-term stability of diagnostic materials, enabling reliable and safe sample storage in laboratory and clinical settings.
Greiner Holding AG provides medical-grade plastic packaging products which include tubes, microtubes and multiwell consumable items. The company provides its services to clinical laboratories, research institutions and diagnostic kit manufacturers through its sterile barrier systems which include customizable formats and automated laboratory workflow integration.
Oliver Healthcare Packaging develops sterile barrier systems together with customized packaging solutions for IVD products. The company develops new contamination control methods which comply with ISO and FDA standards while creating vial, cartridge and tray designs that support quick product development,secure storage and transportation through cold chain and laboratory conditions.
In-Vitro Diagnostics Packaging Market Report Attributes
| Key Takeaway | Details |
|---|---|
| Market Size & Growth | |
| Base Year | 2025 |
| Market Size in 2025 | USD 8.9 Billion |
| Market Size in 2026 | USD 9.2 Billion |
| Forecast Period 2026-2035 CAGR | 5.5% |
| Market Size in 2035 | USD 14.9 Billion |
| Key Market Trends | |
| Drivers | Impact |
| Rising infectious disease diagnostics volume globally | Drives 24% growth by increasing global testing volumes for infectious diseases, requiring higher consumption of sterile, single-use packaging formats such as vials, tubes, and specimen containers to prevent contamination and ensure diagnostic accuracy. |
| Growth in point-of-care testing kits demand | Supports 21% growth through rapid expansion of point-of-care testing kits, which rely on compact, user-friendly, and tamper-evident packaging formats tailored for decentralized healthcare settings including clinics and pharmacies. |
| Cold chain logistics expansion for temperature-sensitive IVDs | Contributes 19% growth as expanding cold chain logistics for temperature-sensitive reagents and biologics increases demand for insulated, high-barrier, and thermally stable packaging solutions across transport and storage stages. |
| Stringent regulatory requirements for sterile packaging | Adds 17% growth by enforcing stringent regulatory standards for sterile and contamination-free packaging, pushing manufacturers to adopt advanced materials, cleanroom packaging, and validated sealing technologies. |
| Increasing home-based diagnostic testing adoption | Accounts for 19% growth with rising adoption of home-based diagnostic testing, driving demand for easy-to-use, leak-proof, and safe disposal packaging formats designed for non-professional users. |
| Pitfalls & Challenges | Impact |
| Supply chain disruptions in medical-grade plastics | Restrains market growth as supply chain disruptions in medical-grade plastics, including polypropylene and polyethylene, create raw material shortages and price volatility. Dependence on limited certified suppliers and geopolitical trade constraints further impact production timelines and cost stability for IVD packaging manufacturers. |
| Complex validation requirements for sterile packaging | Limits growth as sterile packaging requires complex validation processes, including integrity testing, microbial barrier verification, and regulatory documentation. These time-intensive procedures increase development cycles, raise compliance costs, and delay product commercialization in highly regulated diagnostic applications. |
| Opportunities: | Impact |
| Sustainable and recyclable medical packaging innovations | Presents significant growth potential by accelerating adoption of sustainable and recyclable medical packaging solutions, including bio-based polymers and mono-material designs. Increasing regulatory pressure and healthcare sustainability targets are driving innovation in low-waste, environmentally compliant IVD packaging formats. |
| Smart packaging integration for temperature monitoring | Offers opportunity by enabling smart packaging integration with temperature monitoring technologies such as time-temperature indicators and sensor-based labels. These solutions enhance traceability, ensure reagent stability during transit, and support compliance with cold chain requirements in diagnostics distribution. |
| Market Leaders (2025) | |
| Market Leader |
16.5% market share in 2025 |
| Top Players |
Collective market share in 2025 is 55.2% |
| Competitive Edge |
|
| Regional Insights | |
| Largest Market | North America |
| Fastest growing market | Asia Pacific |
| Emerging countries | Canada, China, India, South Korea |
| Future outlook |
|
What are the growth opportunities in this market?
In-Vitro Diagnostics Packaging Industry News
- In November 2025, Greiner Holding AG highlighted PPWR‑ready sustainable packaging solutions at the Future Packaging Day 2025 event, showcasing innovations in monomaterials and recyclable concepts aligned with emerging European regulatory demands.
- In October 2025, Amcor Limited launched AmSecure APET thermoformed trays and rollstock, a next‑generation, cost‑effective and sterilization‑compatible healthcare packaging solution for thermoformed applications. This product improves supply chain efficiency while maintaining clarity and durability needed in medical and pharmaceutical packaging.
- In July 2025, AptarGroup Incorporated acquired the clinical trial materials manufacturing capabilities of Mod3 Pharma, adding cGMP fill‑finish services to its portfolio. This expands Aptar’s technical footprint in drug delivery and potentially secondary packaging, supporting advanced diagnostic and therapeutic product workflows.
The in-vitro diagnostics packaging market research report includes in-depth coverage of the industry with estimates and forecast in terms of revenue (USD Million) from 2022 – 2035 for the following segments:
Market, By Packaging Product Type
- Bottles & vials
- Tubes & microtubes
- Pouches & bags
- Trays & blisters
- Test cartridges & cassettes
- Cartons & boxes
- Others
Market, By Material Type
- Plastics
- Glass
- Paper & paperboard
- Foil laminates
Market, By IVD Product Category
- Reagents & assay kits
- Sample collection & transport Products
- Rapid diagnostic tests (RDTs)
- Molecular diagnostic kits
- Others
Market, By End-User
- IVD manufacturers
- Contract packaging organizations (CPOs)
- Medical device OEMs
The above information is provided for the following regions and countries:
- North America
- U.S.
- Canada
- Europe
- Germany
- UK
- France
- Spain
- Italy
- Netherlands
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- Latin America
- Brazil
- Mexico
- Argentina
- Middle East and Africa
- South Africa
- Saudi Arabia
- UAE
Frequently Asked Question(FAQ) :
What was the market size of the in-vitro diagnostics packaging market in 2025?
The global market was valued at USD 8.9 billion in 2025, driven by rising infectious disease diagnostics and demand for sterile packaging.
What is the projected size of the in-vitro diagnostics packaging market in 2026?
The market is expected to reach USD 9.2 billion in 2026, supported by growth in point-of-care testing and home-based diagnostics.
What is the forecasted value of the in-vitro diagnostics packaging market by 2035?
The market is projected to reach USD 14.9 billion by 2035, expanding at a CAGR of 5.5%, fueled by cold chain logistics expansion and stricter regulatory standards.
Which segment dominates the in-vitro diagnostics packaging industry?
Plastics led the market with a 54% share in 2025, owing to affordability, durability, and compatibility with single-use diagnostic consumables.
Which application segment dominates the in-vitro diagnostics packaging market?
Bottles & vials dominated in 2025, valued at USD 2.8 billion, driven by their extensive use in reagent storage and compatibility with automated dispensing systems.
What was the value of the North America in-vitro diagnostics packaging industry in 2025?
North America held a 35.1% share in 2025, driven by stringent sterility regulations, contamination control, and adoption of smart packaging technologies.
Who are the key players in the in-vitro diagnostics packaging market?
Leading companies include Amcor, AptarGroup, Corning Incorporated, Greiner Holding AG, and Oliver Healthcare Packaging, collectively holding 55.2% share in 2025, supported by innovation in sustainable and sterile packaging solutions.
In-Vitro Diagnostics Packaging Market Scope
Related Reports