Home > Healthcare > Pharmaceuticals > Disease Specific Drugs > Hemophilia Treatment Market
Hemophilia Treatment Market Size By Disease (Hemophilia A, Hemophilia B), By Product (Recombinant Factor Concentrates, Plasma-derived Factor Concentrates, Extended Half-life Products), Patient, Treatment, Therapy, Drug Class, Route of Administration & Forecast, 2022 - 2028
- Report ID: GMI2772
- Published Date: Feb 2022
- Report Format: PDF
Hemophilia Treatment Market Size
Hemophilia Treatment Market size surpassed USD 11.1 billion in 2021 and is projected to register around 5% CAGR between 2022 and 2028. The hemophilia treatment has enhanced significantly over the past few years, owing to the advent of novel products. Several government and non-government organizations are coming forth to promote awareness related to symptoms, diagnosis, and hemophilia treatment.
For instance, the World Federation of Hemophilia (WFH) is actively operating to improve and sustain care for individuals with inherited bleeding disorders worldwide. Such government bodies are working to ensure that patients have access to world-class care and recognition of their condition.

The notable hemophilia prevalence is demanding the high need for quality hemophilia treatment. Hence, numerous nations are emphasizing to establish novel strategies to enhance access to quality care for people living with hemophilia. In Germany, innovative methods have been introduced to improve the monitoring of such ailments and other related conditions. Additionally, in recent years, the awareness related to the importance of early-stage diagnosis and treatment and its potential effect is surging positively across European countries, thereby boosting the demand for a product that will simulate the market revenue significantly.
| Report Attribute | Details |
|---|---|
| Base Year: | 2021 |
| Hemophilia Treatment Market Size in 2021: | USD 11 Billion |
| Forecast Period: | 2022 to 2028 |
| Forecast Period 2022 to 2028 CAGR: | 5% |
| 2028 Value Projection: | USD 16 Billion |
| Historical Data for: | 2017 to 2021 |
| No. of Pages: | 350 |
| Tables, Charts & Figures: | 815 |
| Segments covered: | Disease, Product, Patient, Treatment, Therapy, Drug class, Route of administration, End-use. |
| Growth Drivers: |
|
| Pitfalls & Challenges: |
|
Hemophilia treatment comprises the administration of medications through intravenous or nasal route in order to prevent frequent internal bleeding that may cause arthritis or destruction of the joint, or infection. It is an inherited bleeding disease that may lead to continuous bleeding or oozing after an injury or surgery. In critical conditions, prolonged bleeding occurs after minor trauma or even in the absence of injury.
However, the high cost associated with the hemophilia treatment may adversely affect the market growth. As per the National Hemophilia Foundation, the average cost for hemophilia treatment is around USD 300,000 a year that makes a significant impact on the procedure margins, especially in today's capitated reimbursement environment. The market growth can be hampered in developing countries having comparatively low-income levels attributed to high costs related to clotting factor therapies.
However, the government in certain developing countries is undertaking several initiatives to reduce the cost of the treatment. In addition to this, a lack of awareness in developing and under-developed regions may restrict the industry growth.
The emergence of COVID-19 pandemic slightly impacted the hemophilia treatment market negatively owing to disruption in global supply chain and logistics. The impact of the coronavirus outbreak on the industry peaked during early 2020 wherein, patients suffering from such ailments were at a higher risk of suffering from coronavirus as they were more prone to infectious diseases.
However, intensive efforts were being undertaken by several organizations, hospitals, and hematologists to navigate the COVID-19 public health crisis. For instance, in concern with the outbreak of the coronavirus, the American Society of Hematology provided a forum for the exchange of information that is beneficial, precise, and up to date to assist hematologists. Hence, such changes in practices are expected to help in restoring the treatment volume post-COVID-19 pandemic.
Hemophilia Treatment Market Trends
The increasing prevalence of hemophilia contributing to the rising demand for treatment options is one of the prime factors driving the hemophilia treatment market expansion. Severe patients need a regular hemophilia treatment regimen to maintain enough clotting factors in the bloodstream to prevent bleeds. As per the Centers for Disease Control and Prevention, hemophilia A affects 1 in 5,000 male births each year in the U.S. This considerable prevalence is set to dramatically impact the market, since the number of dependent patients requiring quality care is increasing. Moreover, patients that receive proper care are expected to develop fewer co-morbid conditions and experience a lower risk of severe bleeding. Thus, the continuously increasing patient number has significantly boosted the demand for hemophilia treatment that in turn will uplift the product adoption rate.
Hemophilia Treatment Market Analysis
Based on disease, the hemophilia treatment market is segmented as hemophilia A and hemophilia B. Hemophilia A disease segment dominated the market in 2021 accounting for around 82% of the total revenue share. This high revenue share of the segment is owing to an increased incidence of hypertension among hemophilia A patients, favourable government initiatives for disease control, and surging disease awareness among the population among others.
As per the Centers for Disease Control and Prevention (CDC) recent data, around 400 babies are born with hemophilia A each year in the U.S. This considerable disease incidence is expected to drive the demand for hemophilia A treatment, thereby propelling the segmental revenue positively. The National Hemophilia Foundation (NHF) is actively working to upsurge the awareness pertaining to symptoms, genetics, diagnosis, and treatment of hemophilia A.
Based on product, the hemophilia treatment market is segmented as recombinant factor concentrates, plasma-derived factor concentrates, extended half-life products, and others. The recombinant factor concentrates segment is expected to expand at a CAGR of around 5% from 2022-2028. This significant share of the segment is due to the several advantages delivered by recombinant factor concentrates in hemophilia treatment.
The recombinant factor VIII concentrate provides a technological solution to prolonging the half-life of and decreasing the risk of formation of alloantibodies (inhibitors) against FVIII in treated patients with hemophilia A (HA). All rFVIII products have a better-quality safety profile, as their development process involves virus inactivation procedures similar to those used in plasma-derived factor concentrates.
For patients with severe hemophilia, prophylaxis with FVIII concentrates is the preferred hemophilia treatment to lessen joint bleeding and other types of hemorrhage, thereby enhancing the health-related quality of life of patients. Thus, the above-mentioned factors will stimulate industry landscape.
Based on patient, the market is segmented as pediatric and adult. The adult patient segment exceeded USD 6,898 million in 2021. The prevalence of hemophilia is significantly increasing among the adult population. As per the recent study data, during the period 2012-2018, around 33,000 males were suffering from hemophilia in the U.S. As the age of patients increases, the requirement for quality care to treat this disorder also increases. Thus, several nations are focusing to improve access to hemophilia diagnosis and treatment.
Further, improvement of the global comprehensive care offered by specialized centers is stimulating the adoption of hemophilia treatment, thereby driving the market revenue. Recent advancements in treatment options have significantly improved the life expectancy of adult patients. Thus, numerous advantages coupled with treatment advancements will surge the adoption of the product among adult patients, thereby amplifying the hemophilia treatment industry share.
Based on treatment, the hemophilia treatment market is segmented as prophylaxis and on demand. The prophylaxis segment exceeded USD 7,998 million in 2021. Prophylaxis treatment is considered as the standard of care for treating factor VIII deficiency in pediatric and adult patients. It involves the administration of clotting factor concentrate to prevent bleeding.
This prophylaxis treatment aids in reducing hemophilic arthropathy & disability, decreasing the requirement for orthopedic surgery, thereby delivering improved quality of life. The majority healthcare providers opt for this treatment option attributed to its efficiency and better clinical outcomes. The developments of prophylactic regimens of factor concentrate infusions allowed their administration at home, thereby positively propelling the treatment acceptance. Hence, the above-mentioned factors are expected to facilitate market expansion over the estimation timeframe.
Based on therapy, the market is segmented as factor replacement therapy and non-factor replacement therapy. The non-factor replacement therapy accounted for more than USD 1,648 million in 2021 and is projected to witness over 21.3% CAGR through 2028. This high growth rate is due to the advantages non-factor replacement therapy offers that include reducing treatment burden, increasing the ability to deliver prophylaxis for patients, etc. Limited real-world data and authenticated practical guidance on these recently licensed treatments resulted in increasing their application in hemophilia therapy.
As per the National Center for Biotechnology Information (NCBI), Emicizumab is currently the only licensed nonfactor therapy that helps with venous access issues, frequent bleeds, etc. This therapy is also suggested to prevent bleeds in patients with inhibitors undergoing surgery. Owing to recent advancements, these novel agents have promising hemostatic properties and supports marked reductions of bleeding episodes in hemophilia patients with or without inhibitors.
Thus, the major players in the hemophilia treatment market focuses on developing such advanced therapies with potential advantages comprising marked suppression of the onset of arthropathy. Hence, the increased acceptance of non-factor replacement therapy is predicted to boost the industry expansion.
Based on drug class, the hemophilia treatment market is segmented as vasopressin and coagulation factors. The vasopressin segment accounted for more than USD 573 million in 2021 and is projected to witness 6% CAGR rate till 2028. This considerable segmental share is owing to the improved survival chances in critical patients with the use of vasopressin. As per the National Hemophilia Foundation, this drug can be used for nose & mouth bleeds, joint & muscle bleeds, and before & after surgery among others. It can be administered through both intravenous and nasal routes, thereby reducing the systemic adverse effects of the drugs.
DDAVP (desmopressin acetate) is the synthetic form of vasopressin, a natural antidiuretic hormone that aids stop bleeding due to its clinical efficacy & safety and the availability of concentrated formulation for the intravenous and nasal route of drug administration. As a result, several advantages offered by the use of vasopressin are set to boost the segmental growth.
Based on route of administration, the market is segmented as injectable and nasal spray. The injectables segment is expected to witness over4.9% CAGR between 2022 and 2028. This significant segment growth is due to the fast-acting and efficient mode of drug administration delivered by injectables. This route of administration is recommended for patients that witness challenging circumstances associated with oral intake of medicines.
Intravenous injection route in hemophilia treatment has related to enhancing clinical outcomes, surging the life expectancy, functional status, and quality of life among others. Thus, the majority of the medications and factor concentrate products are intravenously given to patients for improved and quick outcomes.

Based on end-use, the hemophilia treatment market is segmented as hospitals, clinics, hemophilia treatment centers, and others. The hospitals segment accounted for USD 3,382 million in 2021 attributed to the accessibility to board-certified skilled hematologists, the importance of delivering quality care plans, and their improved efficiency among others. These healthcare facilities provide affordable treatment procedures that help both patients and insurers equally.
Easy availability of novel therapies surges the preference for bleeding disorder treatment in hospitals. Also, comprehensive care plans and continuous monitoring offered in hospitals are anticipated to increase the patient preference for these healthcare facilities. Thus, the growing hospital admissions with such inherited bleeding ailments and demand for advanced treatments are projected to fuel the adoption of the product in these facilities.
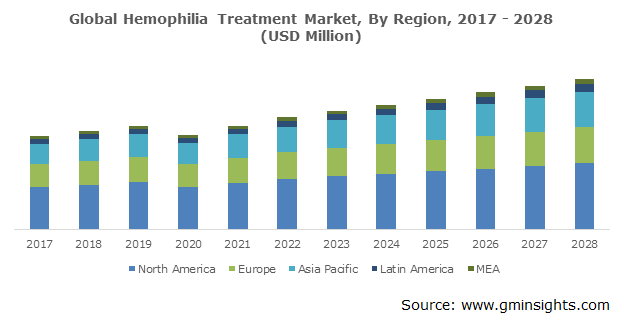
U.S. hemophilia treatment market is poised to surpass USD 6,297 million by 2028. This high growth in the country is owing to several factors including increased government spending on healthcare and R&D activities along with the presence of established manufacturers. Surging awareness related to technologically advanced products and rising target patient pool is also contributing to the business revenue. As per the World Federation of Hemophilia (WFH), in 2018, around 17,757 individuals were diagnosed with hemophilia in the U.S. The organization comprising Hemophilia Federation of America (HFA), commonly delivers continuous treatment for patients by presenting different co-pay initiatives.
Further, in 2021, BioMarin Pharmaceutical Inc. announced a significant Phase 3 gene therapy trial results in adults with severe hemophilia A in the U.S. Such ongoing R&D activities are expected to fuel the market demand over the forecast period. As a result, the above-mentioned factors are anticipated to augment the product adoption in the country.
Hemophilia Treatment Market Share
Some of the major players operating in the hemophilia treatment industry are:
- Bayer Healthcare
- Biogen
- Biotest AG
- CSL Behring
- Ferring B.V
- Genentech
- Kedrion
- Novo Nordisk
- Octapharma
These leading market players are implementing advanced technologies to cater to the large unmet needs of the consumers.
Recent industry developments:
- In February 2019, Novo Nordisk announced the U.S. FDA approval for its biologics license application for Esperoct. This medicine is used in pediatric and adult patients suffering from congenital factor VIII deficiency to lessen the frequency of bleeding episodes. This launch helped the company to introduce innovative products, further offering a competitive edge.
- In August 2018, Bayer Healthcare received the U.S. FDA approval for new extended half-life hemophilia A product for patients 12 years and above. Jivi is a rFVIII replacement therapy that has approved to stop or prevent bleeding. This approval helped the company to strengthen its product portfolio across the globe.
Hemophilia treatment market research report includes an in-depth coverage of the industry with estimates & forecast in terms of revenue in USD from 2017 to 2028, for the following segments:
Click here to Buy Section of this Report
By Disease
- Hemophilia A
- Severe
- Moderate
- Mild
- Hemophilia B
- Severe
- Moderate
- Mild
By Product
- Recombinant Factor Concentrates
- Factor VIII
- Factor IX
- Plasma-derived Factor Concentrates
- Factor VIII
- Factor IX
- Extended Half-life Products
- Factor VIII
- Factor IX
- Others
By Patient
- Pediatric
- 0 to 4
- 5 to 13
- 14 to18
- Adult
- 19 to 44
- 45+
By Treatment
- Prophylaxis
- On Demand
By Therapy
- Factor Replacement Therapy
- Non-factor Replacement Therapy
By Drug Class
- Vasopressin
- Coagulation Factors
By Route of Administration
- Injectable
- Nasal Spray
By End-use
- Hospitals
- Clinics
- Hemophilia Treatment Centers
- Others
The above information is provided for the following regions and countries:
- North America
- U.S.
- Canada
- Europe
- Germany
- UK
- France
- Spain
- Italy
- Asia Pacific
- China
- Japan
- India
- Australia
- South Korea
- Latin America
- Brazil
- Mexico
- Argentina
- Middle East & Africa
- South Africa
- Saudi Arabia
- UAE

